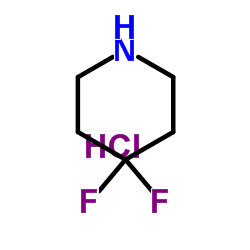
4,4-Difluoropiperidine Hydrochloride
- CAS No.144230-52-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity fluorinated piperidine salt designed for advanced pharmaceutical synthesis and organic research applications with guaranteed quality.
Request Bulk PricingProduct Technical Details
Product Overview
4,4-Difluoropiperidine Hydrochloride represents a critical class of fluorinated heterocyclic compounds essential for modern medicinal chemistry. As a stable hydrochloride salt, this intermediate offers enhanced handling properties and solubility profiles compared to its free base counterpart. The incorporation of fluorine atoms at the 4-position of the piperidine ring significantly influences the metabolic stability and lipophilicity of downstream drug candidates, making it a highly sought-after building block in the development of novel therapeutic agents.
Our manufacturing process ensures consistent quality and high purity levels suitable for rigorous research and development environments. We adhere to strict quality control protocols to guarantee that every batch meets the specified chemical and physical parameters required for complex organic synthesis workflows.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 4,4-Difluoropiperidine Hydrochloride |
| CAS Number | 144230-52-4 |
| Molecular Formula | C5H10ClF2N |
| Molecular Weight | 157.59 g/mol |
| Appearance | Light-Yellow Solid |
| Assay (GC) | ≥99.0% |
| Moisture Content | ≤0.5% |
| Melting Point | 173-177°C |
| Residue on Ignition | ≤0.1% |
Applications and Benefits
This fluorinated intermediate is primarily utilized in the synthesis of pharmaceutical active ingredients where metabolic stability is paramount. The gem-difluoro substitution pattern provides unique electronic effects that can improve binding affinity and bioavailability in final drug products. Key advantages of sourcing this material from our facility include:
- High chemical purity ensuring minimal side reactions during synthesis.
- Stable salt form for easier storage and handling in laboratory settings.
- Consistent batch-to-batch reproducibility for reliable scale-up.
- Comprehensive documentation including Certificates of Analysis (COA).
- Flexible packaging options to suit both research and pilot plant needs.
Quality Assurance and Storage
Quality is the cornerstone of our production philosophy. Each lot undergoes rigorous testing using advanced analytical techniques such as Gas Chromatography (GC) and Nuclear Magnetic Resonance (NMR) to verify identity and purity. We maintain full traceability from raw materials to finished goods. To maintain optimal stability, the product should be stored in a cool, ventilated place away from direct sunlight and moisture. Proper sealing is recommended to prevent hygroscopic absorption which could affect performance in sensitive reactions.
We are committed to supporting global pharmaceutical and chemical industries with reliable supply chains. Our logistics team ensures timely delivery while maintaining product integrity during transit. For specific technical inquiries or custom synthesis requirements, our expert team is available to provide detailed support and documentation.



