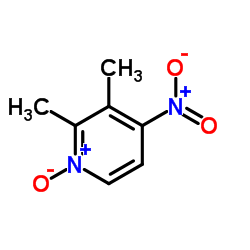
4-Nitro-2,3-lutidine-N-oxide
- CAS No.37699-43-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-Nitro-2,3-lutidine-N-oxide (CAS 37699-43-7) designed for pharmaceutical API synthesis, specifically Lansoprazole and Rabeprazole intermediates.
Request Bulk PricingProduct Technical Details
Product Overview
4-Nitro-2,3-lutidine-N-oxide is a specialized organic compound widely recognized for its critical role in the synthesis of proton pump inhibitors (PPIs). As a key pharmaceutical intermediate, this substance serves as a foundational building block in the manufacturing of high-value active pharmaceutical ingredients (APIs). Our facility produces this compound under strict quality control standards to ensure consistency and reliability for downstream chemical synthesis processes.
The chemical structure features a pyridine N-oxide core with nitro and methyl substitutions, providing unique reactivity profiles essential for complex organic transformations. We supply this material to global pharmaceutical manufacturers and research institutions who require precise molecular specifications for their production lines.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 37699-43-7 |
| Molecular Formula | C7H8N2O3 |
| Molecular Weight | 168.150 |
| Appearance | Yellow crystalline powder |
| Assay (Purity) | ≥98.0% |
| Melting Point | 89-93°C |
| Loss on Drying | ≤0.5% |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 403.3±40.0 °C at 760 mmHg |
Industrial Applications
This intermediate is primarily utilized in the production of gastrointestinal medications. It acts as a crucial precursor in the synthesis pathways for Lansoprazole, Rabeprazole, and Rabeprazole sodium. These final API products are essential treatments for acid-related disorders, making the quality of this intermediate vital for patient safety and drug efficacy.
Our manufacturing process ensures that the impurity profile is minimized, facilitating smoother downstream reactions and higher yields in the final API synthesis. Chemical engineers value this material for its stability and predictable reactivity during nitration and reduction steps.
Quality Assurance and Safety
- Strict adherence to GMP-like guidelines for intermediate production.
- Comprehensive Certificate of Analysis (COA) provided with every batch.
- Regular third-party testing to verify structural integrity and purity levels.
- Robust supply chain management ensuring timely global delivery.
Safety is paramount in handling chemical intermediates. This product should be stored in a cool, ventilated place away from incompatible substances. Personnel must utilize appropriate personal protective equipment (PPE) during handling to prevent inhalation or skin contact. We recommend consulting the Safety Data Sheet (SDS) for detailed hazard information and emergency procedures.
Packaging and Logistics
Standard packaging consists of 25 kg drums, designed to maintain product integrity during transit. However, we understand that different production scales require different solutions. Therefore, we offer flexible packaging options according to specific customer requirements. Our logistics partners are experienced in hazardous material shipping, ensuring compliance with international transport regulations.



