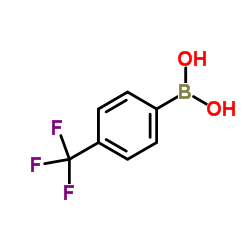
4-Trifluoromethylphenylboronic Acid
- CAS No.128796-39-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-Trifluoromethylphenylboronic acid (CAS 128796-39-4) designed for pharmaceutical intermediate synthesis and cross-coupling reactions.
Request Bulk PricingProduct Technical Details
Product Overview
4-Trifluoromethylphenylboronic Acid is a highly specialized organoboron compound that plays a pivotal role in modern medicinal chemistry and organic synthesis. As a key building block, this substance is extensively utilized in the construction of complex pharmaceutical architectures, particularly where the introduction of a trifluoromethyl group is required to enhance metabolic stability and lipophilicity. Our manufacturing process ensures exceptional consistency and reliability for large-scale industrial applications.
This chemical entity is recognized for its robust performance in palladium-catalyzed cross-coupling reactions. The presence of the electron-withdrawing trifluoromethyl moiety makes it a valuable reagent for synthesizing biaryl structures found in numerous active pharmaceutical ingredients. We maintain strict quality control protocols to guarantee that every batch meets the rigorous demands of global pharmaceutical supply chains.
Technical Specifications
Our production facility adheres to international standards for chemical manufacturing. The following table outlines the critical physical and chemical parameters verified through our quality assurance laboratory. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) to validate compliance with specified limits.
| Parameter | Specification |
|---|---|
| CAS Number | 128796-39-4 |
| Molecular Formula | C7H6BF3O2 |
| Molecular Weight | 189.93 g/mol |
| Appearance | White Crystal Powder |
| Purity (HPLC) | ≥98.0% |
| Water Content | ≤0.50% |
| Melting Point | 245-250 °C |
| Storage Conditions | Cool, Dry, Ventilated |
Industrial Applications
The primary utility of 4-Trifluoromethylphenylboronic Acid lies in its function as a versatile intermediate for drug synthesis. It is notably employed as a critical precursor in the manufacturing of Lomitapide, an important therapeutic agent. Beyond this specific application, the compound facilitates direct cross-coupling reactions with allyl alcohol, enabling the formation of complex molecular frameworks essential for advanced material science and agrochemical development.
Furthermore, this boronic acid derivative supports N-arylation reactions involving imidazole and amines when used with copper exchanged calcium fluorophosphate catalysts. It is also compatible with microwave-assisted cross-coupling protocols using acid chlorides to generate aryl ketones efficiently. These diverse reaction pathways highlight its flexibility as a synthon in multi-step organic synthesis routes.
Quality Assurance and Safety
We prioritize safety and quality in every aspect of our operation. The product is classified under transport hazard class 3 with proper shipping names adhering to ADR/RID regulations for esters. Our packaging options include standard 25kg drums, with customization available to meet specific logistical requirements. Proper storage involves keeping the container tightly closed in a well-ventilated area away from moisture and heat sources to maintain stability.
As a global manufacturer, we are committed to providing reliable supply chains for research and production facilities. Our team supports clients with technical data sheets, safety data sheets, and regulatory documentation to ensure seamless integration into your manufacturing processes. Contact our sales department for detailed information on bulk pricing and availability.



