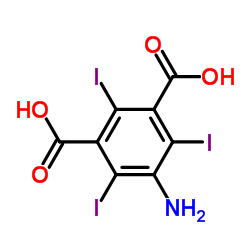
5-Amino-2,4,6-triiodoisophthalic acid
- CAS No.35453-19-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 5-Amino-2,4,6-triiodoisophthalic acid is a critical intermediate for nonionic iodinated X-ray contrast agents. We offer factory-direct pricing with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
5-Amino-2,4,6-triiodoisophthalic acid, frequently referred to by its acronym ATIPA, represents a cornerstone chemical building block in the modern radiopharmaceutical industry. This highly specialized organic compound is characterized by its triiodinated aromatic structure, which provides the necessary radiopacity required for advanced medical imaging applications. As a key precursor, it undergoes further synthetic transformation to produce nonionic iodinated X-ray contrast media, ensuring patient safety and image clarity during diagnostic procedures.
Our manufacturing facility adheres to rigorous international standards to produce this intermediate with exceptional consistency. The chemical stability and high iodine content make it an indispensable raw material for synthesizing prominent contrast agents such as Iohexol, Ioversol, and Iopamidol. We prioritize purity and structural integrity to support downstream pharmaceutical manufacturers in meeting their strict regulatory compliance requirements.
Technical Specifications
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | Light yellow or off-white powder | Conform |
| Purity (HPLC) | ≥98.5% | 99.69% |
| Melting Point | 265-270℃ | 266.6-268.3℃ |
| Loss on drying | ≤1.0% | 0.47% |
| Any other individual impurity | ≤0.5% | Conform |
| Total sum of impurities | ≤1.5% | 0.31% |
Industrial Applications
The primary application of this compound lies within the synthesis of nonionic iodinated X-ray contrast agents. These agents are vital for enhancing the visibility of internal structures in computed tomography (CT) scans and angiography. The high purity level of our 5-Amino-2,4,6-triiodoisophthalic acid ensures that the final pharmaceutical products meet the stringent safety profiles required for intravenous administration.
- Serves as a critical intermediate for Iohexol production.
- Used in the manufacturing process of Ioversol.
- Essential precursor for Iopamidol synthesis.
- Supports the development of next-generation imaging solutions.
Quality Assurance and Packaging
Quality control is paramount in the production of pharmaceutical intermediates. Each batch undergoes comprehensive analysis using High-Performance Liquid Chromatography (HPLC) to verify purity levels exceeding 98.5%. We maintain detailed documentation for every shipment, including Certificates of Analysis (COA) and stability data. Our packaging solutions are designed to protect the chemical integrity during transit, typically utilizing 25 kg barrels, though custom packaging configurations are available to meet specific logistical needs.
For storage, we recommend maintaining a cool, ventilated warehouse environment away from direct heat sources. The temperature should not exceed 37 degrees Celsius to prevent degradation. As a responsible global manufacturer, we ensure all handling procedures align with safety regulations, classifying this material appropriately for transport. Contact our sales team for detailed specifications, bulk pricing structures, and sample requests to support your production planning.



