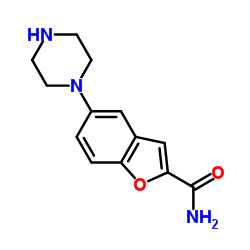
5-Piperazin-1-yl-1-benzofuran-2-carboxamide
- CAS No.183288-46-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity pharmaceutical intermediate used in the synthesis of advanced therapeutic agents. Reliable bulk supply with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
5-Piperazin-1-yl-1-benzofuran-2-carboxamide is a sophisticated organic compound characterized by its benzofuran core linked to a piperazine moiety. This chemical structure serves as a vital building block in the pharmaceutical industry, specifically designed for the construction of complex active pharmaceutical ingredients. Our manufacturing process ensures that every batch meets stringent quality standards required for downstream drug synthesis. The compound is engineered to provide high reactivity and stability, making it an ideal candidate for multi-step organic synthesis routes.
Key Specifications
| Parameter | Value |
|---|---|
| Chemical Name | 5-Piperazin-1-yl-1-benzofuran-2-carboxamide |
| CAS Number | 183288-46-2 |
| Molecular Formula | C13H15N3O2 |
| Molecular Weight | 245.28 g/mol |
| Purity | ≥99.0% |
| Appearance | White or light yellow powder |
| Density | 1.268 g/cm3 |
| Boiling Point | 505.3 °C at 760 mmHg |
Industrial Applications
This intermediate plays a crucial role in the production of modern antidepressant medications. It acts as a key precursor in the synthesis of Vilazodone and related pharmaceutical compounds. The integration of the piperazine ring enhances the biological activity profile of the final drug product. Our clients utilize this material to develop treatments targeting neurological disorders. The high purity level ensures minimal impurity carryover into the final API, which is critical for regulatory compliance and patient safety. We support pharmaceutical companies in scaling their production from clinical trials to commercial manufacturing.
Quality Assurance and Manufacturing
As a global manufacturer, we adhere to rigorous quality control protocols. Each production lot undergoes comprehensive testing using advanced analytical techniques such as HPLC and NMR. We provide a Certificate of Analysis (COA) with every shipment to verify identity and purity. Our facility is equipped to handle large-scale orders while maintaining consistency. We understand the importance of supply chain reliability in the pharmaceutical sector. Our team works closely with clients to customize packaging and delivery schedules according to specific project requirements.
Storage and Handling
To maintain optimal stability, this product should be stored in a cool and dry environment. Containers must be well-closed to protect against moisture and strong light. The recommended storage temperature is in a refrigerator or a controlled room temperature area away from heat sources. Proper handling procedures should be followed to ensure safety during transfer and weighing. We recommend consulting the safety data sheet for detailed handling instructions. Our packaging options include 25kg cardboard drums or custom configurations based on customer needs.



