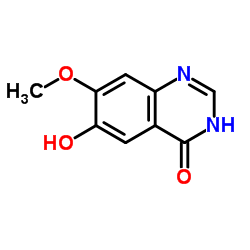
6-Hydroxy-7-methoxy-1H-quinazolin-4-one
- CAS No.179688-52-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of kinase inhibitors. Available in bulk quantities with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
6-Hydroxy-7-methoxy-1H-quinazolin-4-one is a specialized heterocyclic compound serving as a critical building block in modern pharmaceutical synthesis. This quinazolinone derivative is extensively utilized in the development of targeted therapeutic agents, particularly within the realm of tyrosine kinase inhibitors. Our manufacturing process ensures consistent molecular integrity and high chemical purity, making it an ideal choice for research and production-scale applications.
As a key precursor, this compound plays a vital role in constructing complex molecular architectures required for advanced medicinal chemistry. The presence of both hydroxy and methoxy substituents on the quinazolinone scaffold provides unique reactivity profiles, facilitating efficient downstream functionalization. We adhere to strict quality control protocols to guarantee that every batch meets the rigorous demands of pharmaceutical manufacturing.
Technical Specifications
| Item | Specification |
|---|---|
| Chemical Name | 6-Hydroxy-7-methoxy-1H-quinazolin-4-one |
| CAS Number | 179688-52-9 |
| Molecular Formula | C9H8N2O3 |
| Molecular Weight | 192.17 g/mol |
| Appearance | Yellow powder or gray to brown powder |
| Purity | ≥98.0% |
| Loss on Drying | ≤1.0% |
| Residue on Ignition | ≤0.5% |
| Density | 1.47 g/cm3 |
| Boiling Point | 503.3°C at 760 mmHg |
| Flash Point | 258.2°C |
Industrial Applications
This chemical intermediate is primarily employed in the synthesis of specific kinase inhibitors used in oncology research and development. Its structural features allow for versatile coupling reactions, enabling chemists to introduce various side chains necessary for biological activity optimization. The compound is suitable for use in multi-step organic synthesis pathways where high fidelity and minimal impurity profiles are essential.
- Pharmaceutical API synthesis intermediate
- Medicinal chemistry research and development
- Custom synthesis for kinase inhibitor projects
- Quality control standard for related compounds
Quality Assurance and Storage
We implement comprehensive quality assurance measures throughout the production lifecycle. Each batch undergoes rigorous testing using advanced analytical techniques such as HPLC and NMR to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment to ensure full traceability and compliance with industry standards.
For optimal stability, the product should be stored in a cool and dry environment within well-closed containers. It is crucial to protect the material from moisture and strong light or heat sources to prevent degradation. Proper handling procedures should be followed to maintain the chemical integrity during transportation and storage. We offer flexible packaging options, including 25kg cardboard drums, tailored to meet specific customer logistics requirements.



