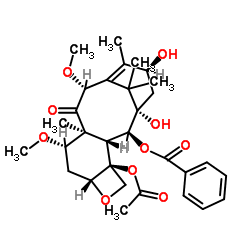
7,10-Dimethoxy-10-DAB
- CAS No.183133-94-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 7,10-Dimethoxy-10-DAB (CAS 183133-94-0) designed for pharmaceutical synthesis. Reliable supply for Cabazitaxel production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
7,10-Dimethoxy-10-DAB, also known by its chemical synonym 7,10-MeO-10-DAB, represents a critical advanced intermediate in the synthesis of complex taxane-based pharmaceutical agents. With the CAS registry number 183133-94-0, this compound serves as a foundational building block for the production of next-generation antineoplastic APIs. Our facility specializes in the large-scale manufacturing of this high-value chemical, ensuring consistent molecular integrity and purity levels that meet rigorous international pharmaceutical standards.
The molecular structure, defined by the formula C31H40O10, requires precise synthetic pathways to maintain stereochemical accuracy. As a key precursor, the quality of this intermediate directly influences the efficacy and safety profile of the final active pharmaceutical ingredient. We employ state-of-the-art purification technologies to remove impurities, guaranteeing a product that supports robust downstream synthesis reactions.
Technical Specifications
Our production process is monitored at every stage to ensure physical and chemical parameters remain within tight tolerances. The following table outlines the standard specifications for our commercial grade material. Custom specifications may be available upon request for specific research or production needs.
| Parameter | Specification |
|---|---|
| CAS Number | 183133-94-0 |
| Molecular Formula | C31H40O10 |
| Molecular Weight | 572.643 g/mol |
| Appearance | Off-white crystalline powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 675.8±55.0 °C at 760 mmHg |
| Flash Point | 213.0±25.0 °C |
Industrial Applications
This compound is primarily utilized as a specialized intermediate in the manufacturing of Cabazitaxel, a potent taxane derivative used in oncology treatments. The chemical properties, including a LogP value of approximately 4.54 and specific polar surface area, make it suitable for complex organic synthesis routes requiring high stability. Pharmaceutical manufacturers rely on this material to streamline their production workflows while maintaining compliance with regulatory guidelines.
Key advantages of sourcing this intermediate from our facility include:
- Consistent batch-to-batch reproducibility for large-scale API production.
- Comprehensive documentation including Certificate of Analysis (COA) and Method Validation.
- Optimized synthesis routes that reduce environmental impact and waste.
- Technical support for process optimization and scale-up activities.
Quality Assurance and Logistics
Quality control is paramount in the supply of pharmaceutical intermediates. Each batch undergoes rigorous testing using HPLC, NMR, and mass spectrometry to verify identity and purity. We adhere to strict storage protocols, recommending that the material be kept in a cool, ventilated environment to preserve stability over time. Packaging is available in 25 kg drums, with flexible options to accommodate specific customer logistics requirements. As a global manufacturer, we ensure secure shipping and timely delivery to support uninterrupted production schedules worldwide.



