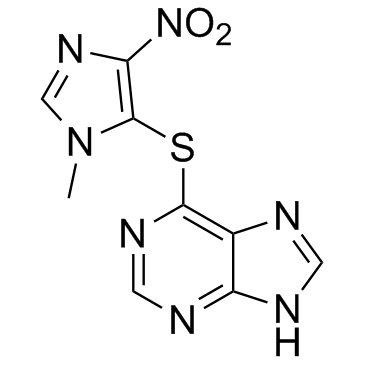
Azathioprine
- CAS No.446-86-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Azathioprine (CAS 446-86-6) supplied for pharmaceutical manufacturing. Meets strict quality standards for immunosuppressive research and development.
Request Bulk PricingProduct Technical Details
Product Overview
Azathioprine is a sophisticated purine analogue antimetabolite widely recognized in the pharmaceutical industry for its significant role in immunosuppressive therapy formulations. As a leading global manufacturer, we provide this critical chemical intermediate with uncompromising quality standards suitable for large-scale pharmaceutical production and advanced research applications. Our supply chain ensures consistent availability of this essential compound for licensed manufacturers developing disease-modifying antirheumatic drugs and other immunosuppressive regimens.
This compound functions as a prodrug that converts to active metabolites within the biological system, interfering with DNA synthesis and thereby modulating immune response. Our manufacturing process adheres to strict international quality management systems, ensuring that every batch meets the rigorous specifications required for pharmaceutical grade active ingredients. We understand the critical nature of supply chain reliability in the pharmaceutical sector and maintain robust inventory levels to support continuous production schedules.
Key Chemical Properties
The chemical stability and physical characteristics of Azathioprine are vital for its processing and formulation into final dosage forms. Our product exhibits consistent physical properties that facilitate efficient handling and integration into various pharmaceutical manufacturing processes. The compound demonstrates high stability under recommended storage conditions, ensuring longevity and performance throughout the supply chain.
- High molecular stability suitable for diverse formulation methods
- Consistent crystalline structure for uniform processing
- Optimized solubility profiles for effective bioavailability
- Rigorous testing for residual solvents and impurities
Quality Specifications
Quality assurance is the cornerstone of our chemical supply operations. Each batch of Azathioprine undergoes comprehensive analytical testing using high-performance liquid chromatography and other advanced methodologies. We provide full documentation including Certificates of Analysis to verify compliance with specified purity levels and physical parameters.
| Parameter | Specification |
|---|---|
| Appearance | Light yellow crystalline powder |
| Purity (HPLC) | Greater than or equal to 98.0 percent |
| Loss on Drying | Less than or equal to 0.5 percent |
| Molecular Formula | C9H7N7O2S |
| Molecular Weight | 277.26 g/mol |
| Melting Point | 243 to 244 degrees Celsius |
Packaging and Storage
Proper packaging and storage are essential to maintain the integrity of pharmaceutical intermediates. We offer flexible packaging solutions designed to protect the product from moisture, light, and contamination during transit and storage. Our standard packaging options accommodate various production scales while ensuring compliance with international shipping regulations for chemical substances.
Standard packaging includes twenty-five kilogram drums with inner liners to ensure product protection. Custom packaging configurations are available upon request to meet specific logistical requirements. Storage recommendations include maintaining a cool and ventilated environment away from direct sunlight and excessive humidity. Adherence to these storage guidelines ensures the chemical remains stable and retains its specified properties throughout its shelf life. Our logistics team coordinates efficient global delivery to support uninterrupted manufacturing operations worldwide.



