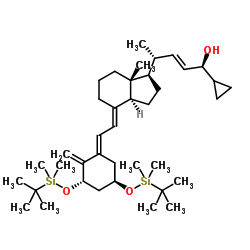
Bis-TBDMS-trans-calcipotriol
- CAS No.112849-27-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
A high-purity pharmaceutical intermediate essential for the synthesis of Calcipotriene, offering reliable stability and consistent quality for large-scale production.
Request Bulk PricingProduct Technical Details
Product Overview
Bis-TBDMS-trans-calcipotriol represents a critical advanced intermediate in the pharmaceutical manufacturing sector, specifically designed for the production of Vitamin D analogs. This sophisticated organic compound features a protected secosterol structure, utilizing tert-butyldimethylsilyl (TBDMS) groups to ensure stability during complex synthetic pathways. As a key precursor, it facilitates the efficient construction of Calcipotriene, a widely recognized active pharmaceutical ingredient used in dermatological therapeutics. Our facility specializes in the precise synthesis of this molecule, adhering to strict international quality standards to support global API manufacturers.
The chemical architecture of this intermediate allows for selective deprotection and functionalization, making it indispensable for multi-step organic synthesis. By employing robust silyl protection strategies, we minimize side reactions and maximize overall yield during the downstream processing of the final active drug substance. This level of molecular precision is vital for maintaining the stereochemical integrity required for biological activity in the final pharmaceutical product.
Technical Specifications
We maintain rigorous control over every batch to ensure compliance with predefined chemical parameters. The following table outlines the standard physical and chemical properties associated with this product grade.
| Parameter | Value |
|---|---|
| CAS Number | 112849-27-1 |
| Molecular Formula | C39H68O3Si2 |
| Molecular Weight | 641.126 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | ≥91.0% |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 628.2±55.0 °C at 760 mmHg |
| Flash Point | 333.7±31.5 °C |
Quality Assurance and Control
Quality is the cornerstone of our manufacturing philosophy. Each production lot undergoes comprehensive analytical testing using high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) spectroscopy. These methods confirm not only the chemical purity but also the structural correctness of the TBDMS protected groups. We provide a detailed Certificate of Analysis (COA) with every shipment, ensuring full traceability and transparency for our B2B partners. Our quality management system is designed to meet the demanding requirements of pharmaceutical supply chains, reducing the risk of production delays due to material inconsistencies.
Industrial Applications
The primary application of Bis-TBDMS-trans-calcipotriol lies in the synthesis of Calcipotriene (CAS 112828-00-9). It serves as a protected intermediate that allows chemists to navigate complex reaction conditions without compromising the sensitive hydroxyl functionalities on the steroid backbone. This capability is crucial for scaling up from laboratory research to industrial manufacturing. Pharmaceutical companies rely on this intermediate to produce consistent batches of topical treatments for psoriasis and other skin conditions. Our product supports both generic drug manufacturing and innovative research projects aimed at developing next-generation Vitamin D therapeutics.
Storage and Handling
To preserve the chemical stability and integrity of this intermediate, proper storage conditions are essential. The material should be kept in a cool, ventilated environment away from direct sunlight and moisture. Standard packaging involves 25 kg drums, though we offer flexible customization to meet specific logistical requirements. Safety data sheets (SDS) are available upon request to ensure safe handling during transport and warehouse management. By choosing our supply, you gain access to a reliable partner committed to safety, compliance, and technical excellence in the fine chemicals industry.



