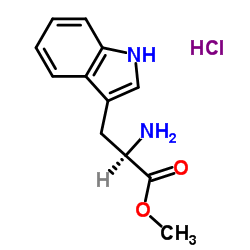
D-Tryptophan Methyl Ester Hydrochloride
- CAS No.14907-27-8
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity D-Tryptophan methyl ester hydrochloride (CAS 14907-27-8) designed for pharmaceutical synthesis. Ideal intermediate for PDE5 inhibitor production with strict quality control.
Request Bulk PricingProduct Technical Details
Product Overview
D-Tryptophan Methyl Ester Hydrochloride is a specialized chiral pharmaceutical intermediate engineered for advanced organic synthesis applications. As a key building block in the production of complex therapeutic agents, this compound offers exceptional stereochemical integrity and reactivity. Our manufacturing process ensures consistent quality suitable for large-scale pharmaceutical development and research initiatives.
This amino acid derivative features a protected indole side chain and a methyl ester functionality, making it highly versatile for peptide coupling and heterocyclic construction. The hydrochloride salt form enhances stability and solubility in various reaction media, facilitating smoother downstream processing in multi-step synthesis routes.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 14907-27-8 |
| Molecular Formula | C12H15ClN2O2 |
| Molecular Weight | 254.713 g/mol |
| Appearance | Off-white powder |
| Assay | ≥98.0% |
| Melting Point | 213-216°C |
| Boiling Point | 390.6°C at 760mmHg |
| Flash Point | 190°C |
Industrial Applications
Primarily utilized as a critical intermediate in the synthesis of phosphodiesterase type 5 (PDE5) inhibitors, this compound plays a vital role in cardiovascular and urological therapeutic development. Its high optical purity ensures the final active pharmaceutical ingredients meet stringent regulatory requirements for chiral drugs.
Beyond specific drug synthesis, D-Tryptophan Methyl Ester Hydrochloride serves as a valuable reagent in medicinal chemistry laboratories for constructing indole-based scaffolds. Researchers leverage its reactivity for developing novel compounds targeting various biological pathways, supporting innovation in drug discovery pipelines.
Quality Assurance and Handling
We adhere to rigorous quality management systems to guarantee product consistency. Each batch undergoes comprehensive analytical testing, including HPLC, NMR, and melting point analysis, to verify identity and purity. A Certificate of Analysis (COA) is provided with every shipment to ensure transparency and compliance with international standards.
- Stored in a cool, ventilated environment to maintain stability.
- Packaged in 25 kg drums or customized according to client requirements.
- Handled under strict safety protocols to prevent contamination.
Our global supply chain ensures reliable delivery for both pilot-scale projects and commercial manufacturing needs. Partner with us for dependable access to high-quality pharmaceutical intermediates that drive your synthesis success.



