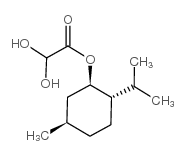
L-Menthyl glyoxylate hydrate
- CAS No.111969-64-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity L-Menthyl glyoxylate hydrate intermediate for pharmaceutical synthesis. Reliable global supply with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
L-Menthyl glyoxylate hydrate is a specialized chiral chemical intermediate widely utilized in the field of advanced organic synthesis. As a key building block, this compound plays a critical role in the construction of complex molecular structures required for pharmaceutical research and development. Our facility produces this material under strict quality control standards to ensure consistency and reliability for downstream manufacturing processes.
The chemical structure features a menthyl ester group attached to a glyoxylic acid moiety, providing unique reactivity profiles suitable for various synthetic routes. The hydrate form ensures stability during storage and handling, making it a preferred choice for industrial-scale applications where reproducibility is paramount.
Chemical Properties and Specifications
This compound is characterized by its distinct physical properties, which are essential for process engineers and chemists during formulation and reaction planning. The material typically presents as a crystalline powder ranging from white to light yellow in color. It possesses a defined melting point and density that facilitate accurate dosing and integration into synthesis protocols.
| Parameter | Specification |
|---|---|
| CAS Number | 111969-64-3 |
| Molecular Formula | C12H22O4 |
| Molecular Weight | 230.30 g/mol |
| Appearance | White to light yellow crystalline powder |
| Assay (GC) | ≥98.0% |
| Melting Point | 76-78°C |
| Density | 1.09 g/cm3 |
| Water Content (KF) | ≤8.50% |
Quality Assurance and Industrial Applications
Quality is the cornerstone of our manufacturing process. Every batch of L-Menthyl glyoxylate hydrate undergoes rigorous testing using advanced analytical techniques such as Gas Chromatography (GC) and Karl Fischer titration. We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing identification, assay results, and impurity profiles to ensure compliance with international standards.
In terms of applications, this intermediate is primarily employed in the synthesis of advanced pharmaceutical compounds. It serves as a vital precursor in multi-step synthesis routes where chirality and functional group compatibility are crucial. Our clients utilize this material for research purposes and the development of therapeutic intermediates, benefiting from its high purity and consistent performance.
- High chemical purity ensures minimal side reactions.
- Stable hydrate form facilitates easier handling and storage.
- Verified chiral integrity for stereoselective synthesis.
- Scalable production capacity to meet bulk demands.
Packaging, Storage, and Logistics
To maintain product integrity during transit, we utilize robust packaging solutions. The standard packaging configuration is 25 kg per drum, designed to protect the material from moisture and physical damage. Customized packaging options are available upon request to suit specific logistical requirements or production line configurations.
For optimal storage, the product should be kept in a cool, ventilated environment away from direct sunlight and sources of ignition. Proper storage conditions help preserve the chemical stability and extend the shelf life of the material. Our logistics team ensures timely global delivery, adhering to all relevant safety regulations for chemical transport. We are committed to supporting our partners with reliable supply chains and technical documentation.



