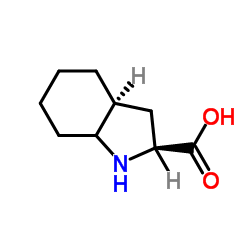
Octahydro-1H-indole-2-carboxylic acid
- CAS No.80828-13-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Octahydro-1H-indole-2-carboxylic acid (CAS 80828-13-3) designed for pharmaceutical synthesis. Ideal intermediate for Trandolapril production with ≥98.0% assay guarantee.
Request Bulk PricingProduct Technical Details
Product Overview
Octahydro-1H-indole-2-carboxylic acid is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Identified by CAS number 80828-13-3, this substance plays a pivotal role in the synthesis of advanced cardiovascular medications. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for downstream manufacturing processes.
As a saturated indole derivative, this carboxylic acid exhibits stable chemical properties suitable for complex multi-step synthesis routes. It is primarily utilized in the production of Angiotensin-Converting Enzyme (ACE) inhibitors, specifically acting as a key precursor for Trandolapril. The structural integrity of this molecule is essential for maintaining the efficacy of the final therapeutic product.
Quality Specifications
We adhere to rigorous industry standards to deliver materials that meet the demanding requirements of pharmaceutical manufacturers. Each batch undergoes comprehensive analytical testing to verify identity, purity, and physical characteristics. The following table outlines the standard specifications for our commercial grade material:
| Parameter | Specification |
|---|---|
| Chemical Name | Octahydro-1H-indole-2-carboxylic acid |
| CAS Number | 80828-13-3 |
| Molecular Formula | C9H15NO2 |
| Molecular Weight | 169.221 |
| Appearance | White to off-white crystalline powder |
| Assay | ≥98.0% |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 318.6±25.0 °C at 760 mmHg |
| Flash Point | 146.5±23.2 °C |
| Refractive Index | 1.508 |
Industrial Applications
The primary application of this compound lies within the realm of pharmaceutical intermediates. It is extensively used in the manufacturing of antihypertensive agents. The high purity level ensures minimal impurity carryover into the final active pharmaceutical ingredient (API), thereby simplifying purification steps and improving overall yield.
- Synthesis of Trandolapril and related ACE inhibitors
- Development of cardiovascular therapeutic agents
- Research and development in organic synthesis
- Custom pharmaceutical manufacturing projects
Production and Logistics
Our manufacturing process is designed to maximize efficiency while maintaining environmental safety standards. We offer flexible packaging solutions to accommodate various production scales, from pilot batches to full commercial runs. Standard packaging consists of 25 kg drums, though custom configurations are available upon request to suit specific logistical needs.
Proper storage is crucial to maintain product stability. We recommend storing the material in a cool, ventilated area away from direct sunlight and moisture. Our global supply chain ensures timely delivery to partners worldwide, supported by comprehensive documentation including Certificates of Analysis (COA) and safety data sheets.



