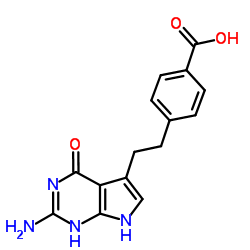
Pemetrexed acid
- CAS No.137281-39-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Pemetrexed Acid is a high-purity pharmaceutical intermediate used in the synthesis of Pemetrexed disodium. We offer reliable bulk supply with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Pemetrexed Acid serves as a pivotal chemical entity in the modern pharmaceutical landscape, specifically within the realm of oncology therapeutics. As the free acid form of the widely recognized antineoplastic agent, this compound acts as a critical precursor in the multi-step synthesis of Pemetrexed disodium. Our manufacturing process ensures that every batch meets rigorous international standards, providing pharmaceutical companies with a reliable foundation for API production. The molecular structure, characterized by a pyrrolo[2,3-d]pyrimidine core linked to a benzoic acid moiety, requires precise handling to maintain stability and purity.
We understand the critical nature of supply chain consistency in the pharmaceutical industry. Our facility is equipped to handle large-scale production demands while maintaining strict quality control protocols. This intermediate is designed for use by licensed pharmaceutical manufacturers engaged in the development and production of antifolate-based chemotherapy agents. By choosing our product, partners gain access to a material that supports regulatory filings and ensures batch-to-batch reproducibility.
Technical Specifications
| Parameter | Specification |
|---|---|
| Appearance | Off-white to light pink powder |
| Purity (HPLC) | ≥98.0% |
| Water Content (K.F) | <0.5% |
| Sulphate Ash | <0.1% |
| Molecular Formula | C15H14N4O3 |
| Molecular Weight | 298.30 g/mol |
| CAS Number | 137281-39-1 |
Quality Control and Impurity Profile
The control of related substances is paramount in the production of oncology intermediates. Our analytical data demonstrates tight control over specific impurities, including Impurities A through F. Each unknown impurity is monitored to ensure it remains within safe thresholds, typically below 0.2% for unspecified variants. The total impurity profile is maintained under 1.0%, which is essential for downstream processing into the final active pharmaceutical ingredient. Heavy metals are controlled to less than 20ppm, and residual solvents such as methanol, ethanol, and acetonitrile are monitored to comply with ICH guidelines.
Our quality assurance team performs comprehensive testing on every lot. This includes assay verification, where the content is typically found between 98% and 102% calculated on an anhydrous basis. The consistency of these results allows our clients to streamline their own validation processes. We provide a Certificate of Analysis (COA) with every shipment, detailing the specific test results for appearance, related substances, moisture, and assay values.
Storage and Handling
Due to its chemical nature, Pemetrexed Acid requires specific storage conditions to prevent degradation. The material should be stored in a cool and dry environment, kept in well-closed containers to protect against moisture absorption. Exposure to strong light or heat should be avoided as these factors can influence the stability of the pyrrolopyrimidine ring system. Standard industrial safety practices should be followed during handling, including the use of appropriate personal protective equipment to minimize exposure.
Industrial Applications
This intermediate is primarily utilized in the manufacturing of injectable chemotherapy formulations. It serves as the key building block for the disodium salt form used in clinical settings. Beyond direct API synthesis, it is also valuable in research and development contexts where new analogues or formulation strategies are being explored. Our global logistics network ensures timely delivery to manufacturing sites worldwide, supporting continuous production schedules without interruption.



