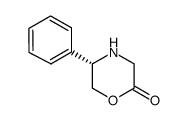
(S)-5-Phenylmorpholin-2-one
- CAS No.144896-92-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of Eliglustat. Available in bulk quantities with strict quality control and comprehensive documentation.
Request Bulk PricingProduct Technical Details
Product Overview
(S)-5-Phenylmorpholin-2-one is a specialized chiral building block extensively utilized in the pharmaceutical industry. This compound serves as a critical intermediate in the synthetic pathway of Eliglustat, a targeted therapy used for the long-term treatment of Gaucher disease type 1 in adults. Our manufacturing process ensures high stereochemical purity, which is essential for the efficacy and safety of the final active pharmaceutical ingredient.
As a dedicated chemical manufacturer, we understand the stringent requirements of modern drug synthesis. The morpholinone structure provides a robust framework for further functionalization, making it a valuable asset for medicinal chemists and process development teams. We commit to delivering consistent quality across all batches to support seamless scale-up from clinical trials to commercial production.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 144896-92-4 |
| Molecular Formula | C10H11NO2 |
| Molecular Weight | 177.20 g/mol |
| Appearance | White powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.149 g/cm3 |
| Boiling Point | 364.3°C at 760 mmHg |
| Flash Point | 174.1°C |
Industrial Applications
The primary application of (S)-5-Phenylmorpholin-2-one lies in the synthesis of glucosylceramide synthase inhibitors. Its specific stereochemistry at the 5-position is crucial for the biological activity of the downstream API. Beyond this specific use, the compound demonstrates versatility in organic synthesis where chiral morpholine derivatives are required.
- Key intermediate for Eliglustat manufacturing
- Utilized in asymmetric synthesis protocols
- Suitable for process research and development
- Compatible with various downstream coupling reactions
Quality Assurance and Storage
Quality control is paramount in our production facility. Each batch undergoes rigorous testing using advanced analytical techniques such as HPLC and NMR to verify identity and purity. We provide a comprehensive Certificate of Analysis (COA) with every shipment to ensure transparency and compliance with international standards.
To maintain stability, the product should be stored in a cool, ventilated area away from direct sunlight and moisture. We offer flexible packaging options, including standard 25 kg drums, and can accommodate custom packaging requirements based on client logistics needs. Our global supply chain ensures timely delivery to support your manufacturing schedules.



