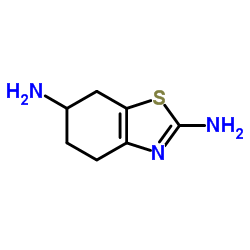
(S)-N-Despropyl Pramipexole
- CAS No.106092-09-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity (S)-N-Despropyl Pramipexole intermediate for pharmaceutical synthesis. Certified quality with full COA available for global B2B partners.
Request Bulk PricingProduct Technical Details
Product Overview
(S)-N-Despropyl Pramipexole is a specialized chiral chemical intermediate primarily utilized in the pharmaceutical industry for the synthesis of dopamine agonist therapeutics. As a key structural analog, this compound serves as a critical building block in the manufacturing process of advanced neurological medications. Our facility ensures that every batch meets rigorous international standards for purity and stereochemical integrity, making it an ideal choice for research and development teams as well as large-scale production units.
This compound is characterized by its specific optical rotation and high chemical stability, which are essential for maintaining the efficacy of the final pharmaceutical product. We understand the complexities of chiral synthesis and provide material that minimizes impurities, thereby streamlining downstream processing and ensuring consistent quality in the final API production.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 106092-09-5 |
| Molecular Formula | C7H11N3S |
| Molecular Weight | 169.25 g/mol |
| Appearance | White or off-white powder |
| Purity | ≥99.0% |
| Specific Optical Rotation | -96° to -120° |
| Melting Point | 222-224°C |
| Loss on Drying | ≤0.1% |
| Residue on Ignition | ≤0.5% |
| Heavy Metals | ≤0.002% |
Industrial Applications
The primary application of (S)-N-Despropyl Pramipexole lies in its role as a precursor for pharmaceutical active ingredients targeting the central nervous system. It is extensively used in the synthesis route of dopamine autoreceptor agonists, which are vital for treating conditions such as Parkinson's disease and Restless Legs Syndrome. Due to its high stereochemical purity, it is also valued in analytical laboratories as a reference standard for quality control testing of finished drug products.
Our clients utilize this intermediate to optimize their manufacturing processes, ensuring that the final drug substance meets all regulatory requirements for chiral purity. Whether for pilot-scale studies or commercial production, this chemical provides a reliable foundation for complex organic synthesis workflows.
Quality Assurance and Safety
We adhere to strict quality management systems to guarantee the consistency and safety of our chemical products. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) detailing test results from HPLC, NMR, and optical rotation measurements. Our packaging ensures protection against moisture and light, preserving the chemical integrity during transit.
- Stored in tightly closed containers to prevent contamination.
- Kept in a cool, dry, and well-ventilated area away from incompatible substances.
- Handled according to standard industrial safety protocols for fine chemicals.
For global partners, we offer flexible packing options, including 25kg cardboard drums or customized solutions based on specific logistical requirements. Our commitment to compliance and quality makes us a trusted supplier for the global pharmaceutical supply chain.



