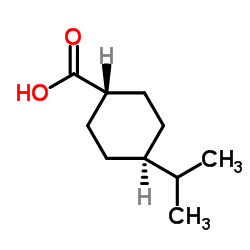
Trans-4-Isopropylcyclohexane Carboxylic Acid
- CAS No.7077-05-6
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Trans-4-Isopropylcyclohexane Carboxylic Acid (CAS 7077-05-6) is a critical pharmaceutical intermediate for Nateglinide synthesis, offering superior isomeric purity and global supply reliability.
Request Bulk PricingProduct Technical Details
Product Overview
Trans-4-Isopropylcyclohexane Carboxylic Acid represents a high-value pharmaceutical intermediate essential for the synthesis of advanced therapeutic agents. Specifically engineered for precision organic synthesis, this compound serves as a critical building block in the production of Nateglinide, a prominent hypoglycemic agent used in diabetes management. Our manufacturing process prioritizes stereochemical purity, ensuring that the trans-isomer predominates while minimizing cis-isomer contamination to meet stringent pharmacopeial standards.
As a leading global manufacturer, we understand the complexities of modern drug development. The structural integrity of this carboxylic acid derivative is paramount for downstream reaction efficiency. We employ advanced chromatographic purification techniques to achieve assay levels exceeding 99.30%, thereby reducing purification burdens for our clients during subsequent synthetic steps. This commitment to quality ensures consistent batch-to-batch reproducibility, a vital factor for regulatory compliance in pharmaceutical manufacturing.
Technical Specifications
Our quality control laboratory utilizes state-of-the-art Gas Chromatography (GC) and Karl Fischer titration to verify every parameter. The following table outlines the typical physical and chemical properties of our premium grade material:
| Parameter | Specification |
|---|---|
| CAS Number | 7077-05-6 |
| Molecular Formula | C10H18O2 |
| Molecular Weight | 170.25 g/mol |
| Appearance | White Crystalline Powder |
| Assay (GC) | ≥ 99.30% |
| Cis Isomer Content | ≤ 0.20% |
| Single Impurity | ≤ 0.30% |
| Melting Point | 92~97℃ |
| Water Content (K.F) | ≤ 0.50% |
| Burning Residue | ≤ 0.20% |
Industrial Applications
This specialized carboxylic acid is primarily utilized within the pharmaceutical sector as a key precursor. Its high stereochemical purity makes it indispensable for the efficient synthesis of insulin secretagogues. Beyond its primary use, the compound's stability and reactivity profile support various organic transformations requiring a robust cyclohexane scaffold. Researchers and process chemists value this material for its predictable behavior in amidation and coupling reactions.
- Critical intermediate for antidiabetic pharmaceutical synthesis.
- High trans-selectivity ensures optimal downstream yield.
- Low impurity profile reduces waste treatment costs.
- Suitable for scale-up from pilot plant to commercial production.
Quality Assurance and Compliance
We adhere to strict quality management systems to guarantee product safety and consistency. Every batch undergoes rigorous testing against internal specifications and customer requirements. Documentation packages are comprehensive, including Certificate of Analysis (COA), Method of Analysis (MOA), and Stability Data. Our facility maintains excellent safety standards, ensuring that all chemical handling procedures comply with international environmental and occupational health regulations. Clients can rely on our transparent supply chain and audit-ready documentation for their regulatory submissions.
Storage and Handling
To maintain optimal quality, Trans-4-Isopropylcyclohexane Carboxylic Acid should be stored in a cool, dry, and well-ventilated facility. Containers must remain tightly sealed to prevent moisture absorption, which could affect the water content specifications. We offer flexible packaging solutions, typically 25kg drums, with customization available to suit specific logistical requirements. Our supply chain is optimized for global delivery, ensuring timely arrival with full documentation. For large-scale projects, we provide dedicated technical support to assist with process optimization and integration into your existing manufacturing workflows.



