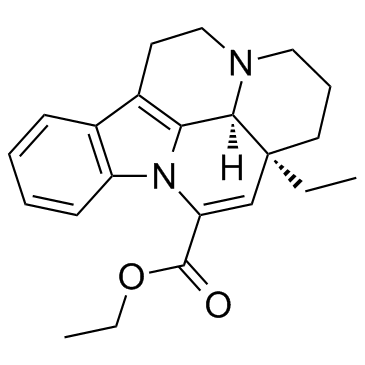
Vinpocetine
- CAS No.42971-09-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Vinpocetine powder for pharmaceutical and nutraceutical formulations, supporting cerebral vascular health applications.
Request Bulk PricingProduct Technical Details
Product Overview
Vinpocetine is a sophisticated semi-synthetic derivative derived from vincamine, a minor alkaloid found in the periwinkle plant. As a leading chemical supplier, we provide high-quality Vinpocetine bulk material designed for integration into complex pharmaceutical and nutraceutical formulations. This compound is widely recognized in the industry for its specific chemical stability and bioavailability profile, making it a preferred choice for manufacturers developing products focused on cerebral health support.
Our manufacturing process adheres to strict quality control standards, ensuring that each batch meets rigorous purity specifications. The material is synthesized through advanced organic chemistry techniques to guarantee consistency in molecular structure and performance. Whether utilized in research settings or large-scale production lines, our Vinpocetine serves as a reliable foundational component for downstream applications.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 42971-09-5 |
| Molecular Formula | C22H26N2O2 |
| Molecular Weight | 350.454 |
| Appearance | White or off-white crystalline powder |
| Assay | ≥99.0% |
| Melting Point | 147-153°C dec |
| Water Content | ≤0.50% |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 419.5±45.0 °C at 760 mmHg |
Industrial Applications
In the B2B chemical sector, Vinpocetine is primarily utilized as a key active ingredient for formulations targeting neurological and vascular health. It is extensively employed in the development of pharmaceutical preparations aimed at supporting cerebral circulation. Additionally, the nutraceutical industry leverages this compound for dietary supplements designed to promote cognitive function and overall brain health.
Our clients value this material for its compatibility with various delivery systems, including tablets, capsules, and liquid formulations. The high purity level ensures minimal interference with other excipients, allowing for precise dosage control and enhanced product efficacy. We support our partners with technical data and formulation guidance to streamline their production processes.
Quality Assurance and Packaging
Quality is paramount in our supply chain. Every shipment of Vinpocetine is accompanied by a comprehensive Certificate of Analysis (COA), verifying identity, purity, and physical characteristics. We employ robust packaging solutions to maintain product integrity during transit. Standard packaging includes 25kg drums, though we offer flexible customization to meet specific logistical requirements.
Proper storage is essential to maintain the chemical stability of the product. We recommend storing the material in a cool, dry, and well-ventilated environment. Containers should remain tightly closed when not in use to prevent moisture absorption and contamination. Our logistics team ensures global shipping compliance, delivering safe and timely supplies to manufacturers worldwide.



