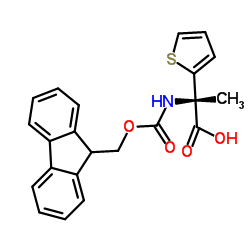
Fmoc-3-L-Ala(2-Thienyl)-OH
- CAS No.130309-35-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Fmoc-3-L-Ala(2-Thienyl)-OH is a specialized protected amino acid derivative designed for efficient peptide synthesis. Manufactured under strict quality controls with ≥98% purity.
Request Bulk PricingProduct Technical Details
Product Overview
Fmoc-3-L-Ala(2-Thienyl)-OH is a premium protected amino acid derivative extensively utilized in the field of organic synthesis and pharmaceutical research. As a crucial building block, this compound features a fluorenylmethoxycarbonyl (Fmoc) protecting group attached to a thienyl-substituted alanine structure. This specific configuration provides enhanced stability and reactivity profiles essential for complex peptide chain assembly. Our manufacturing process ensures consistent quality, making it an ideal choice for laboratories and industrial facilities focused on advanced medicinal chemistry.
The compound is characterized by its high optical purity and minimal presence of D-enantiomers, which is critical for maintaining the biological activity of synthesized peptides. Produced under stringent Good Manufacturing Practices (GMP), each batch undergoes rigorous analytical testing to confirm identity, purity, and structural integrity. This commitment to quality assurance supports researchers in achieving reproducible results during drug discovery and development phases.
Key Specifications
| Parameter | Specification | Typical Result |
|---|---|---|
| Appearance | White to off-white powder | Conform |
| Purity (HPLC) | ≥98.0% | 99.48% |
| Optical Purity | ≤0.5% D-enantiomer | ND |
| Melting Point | 165-178℃ | 169.9-175.0℃ |
| Specific Rotation | -32°±2° (C=1 in DMF) | -32.8° |
| Water Content | ≤1.0% | 0.08% |
| Loss on Drying | ≤1.0% | 0.34% |
Industrial Applications
This specialized amino acid derivative serves as a fundamental component in Solid-Phase Peptide Synthesis (SPPS). The Fmoc protection strategy allows for mild deprotection conditions, preserving sensitive functional groups within the peptide sequence. The incorporation of the 2-thienyl group introduces unique steric and electronic properties, potentially enhancing the metabolic stability or binding affinity of the final peptide therapeutic.
- Utilized in the synthesis of novel peptide-based pharmaceuticals.
- Employed in the development of diagnostic agents and biomarkers.
- Supports research into protein-protein interaction inhibitors.
- Compatible with automated peptide synthesizers for high-throughput production.
Storage and Handling
To maintain optimal stability and performance, Fmoc-3-L-Ala(2-Thienyl)-OH should be stored in a cool, dry environment at room temperature. The container must be kept tightly sealed to prevent exposure to moisture and light, which could degrade the Fmoc group over time. When handling this material, standard laboratory safety protocols should be followed, including the use of appropriate personal protective equipment. For long-term storage, ensuring low humidity conditions is recommended to prevent clumping or hydrolysis. Comprehensive Certificates of Analysis (COA) are provided with every shipment to verify compliance with stated specifications.



