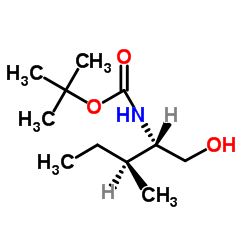
N-Boc-(2S,3S)-(-)-2-Amino-3-methyl-1-pentanol
- CAS No.106946-74-1
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity chiral building block for peptide synthesis. Reliable supply for pharmaceutical intermediate production with full documentation.
Request a Quote & Full COAProduct Technical Details
Product Overview
N-Boc-(2S,3S)-(-)-2-Amino-3-methyl-1-pentanol is a specialized chiral building block extensively utilized in the synthesis of complex pharmaceutical compounds and peptide therapeutics. As a protected amino alcohol derivative, this compound features a tert-butyloxycarbonyl (Boc) group that ensures stability during multi-step organic transformations. The specific (2S,3S) stereochemistry is critical for maintaining the biological activity and structural integrity of the final active pharmaceutical ingredients (APIs).
Our manufacturing process adheres to strict quality control protocols to ensure consistent optical purity and chemical integrity. This material serves as a vital synthon in asymmetric synthesis, enabling chemists to construct intricate molecular architectures with high precision. The robust protection strategy offered by the Boc group allows for selective deprotection under mild acidic conditions, facilitating versatile downstream processing.
Technical Specifications
| Parameter | Value |
|---|---|
| CAS Number | 106946-74-1 |
| Molecular Formula | C11H23NO3 |
| Molecular Weight | 217.31 g/mol |
| Purity | ≥99.0% |
| Appearance | White powder |
| Density | 0.984 g/cm3 |
| Boiling Point | 121-126 °C at 0.6 mm Hg |
| Refractive Index | n20/D 1.455 |
Industrial Applications
This chiral intermediate is primarily employed in the pharmaceutical industry for the development of peptidomimetics and protease inhibitors. Its high optical purity makes it an ideal candidate for constructing stereodefined backbones in drug discovery programs. Additionally, it finds application in the synthesis of specialized ligands for asymmetric catalysis, where chirality transfer is essential for reaction outcomes.
- Peptide synthesis and modification
- Pharmaceutical intermediate production
- Asymmetric catalysis research
- Development of chiral auxiliaries
Storage and Handling
To maintain optimal stability, this product should be stored in a cool, dry environment at approximately 0°C. The container must remain tightly closed to prevent moisture absorption, which could compromise the integrity of the Boc protecting group. Handling should be conducted under inert atmosphere conditions when possible to minimize exposure to atmospheric contaminants. Proper personal protective equipment is recommended during transfer and weighing operations.
Quality Assurance
Every batch undergoes rigorous analytical testing including HPLC, NMR, and optical rotation measurements to verify identity and purity. We provide comprehensive documentation such as Certificates of Analysis (COA) and Method Validation Reports with each shipment. Our commitment to quality ensures that researchers and production teams receive materials that meet the highest industry standards for consistency and reliability.