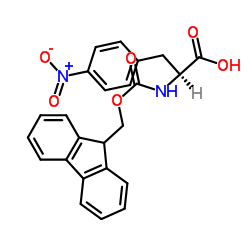
N-Fmoc-4-Nitro-L-Phenylalanine
- CAS No.95753-55-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium grade N-Fmoc-4-Nitro-L-Phenylalanine (CAS 95753-55-2) designed for high-efficiency peptide synthesis. Available in bulk with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
N-Fmoc-4-Nitro-L-Phenylalanine is a highly specialized protected amino acid derivative essential for modern solid-phase peptide synthesis (SPPS). As a critical building block, this compound features a fluorenylmethoxycarbonyl (Fmoc) protecting group on the alpha-amino function and a nitro group at the para-position of the phenyl ring. This specific chemical configuration allows chemists to introduce unique electronic and steric properties into peptide chains, facilitating the development of novel pharmaceutical intermediates and bioactive molecules.
Our manufacturing process ensures exceptional stability and reactivity, making it an ideal choice for complex peptide assembly. The compound is rigorously tested to meet international standards for pharmaceutical research and development. Whether used in the synthesis of integrin antagonists or marine peptide analogs, this reagent provides the reliability required for high-stakes laboratory environments.
Key Specifications
We maintain strict quality control protocols to guarantee consistency across every batch. The following table outlines the critical physical and chemical parameters verified during our analysis:
| Items of Analysis | Standard | Test Results |
|---|---|---|
| Appearance | White to off-white powder | Conform |
| Purity (HPLC) | ≥98.0% | 99.44% |
| Melting Point | 200-220°C | 209.4-210.8°C |
| Specific Rotation | -40°±3° (C=1 in DMF) | -38.2° |
| Clarity of Solution | Clear in DMF | Conform |
| Loss on Drying | ≤2.0% | 0.32% |
| Water Content | ≤2.0% | 0.41% |
Industrial Applications
This protected amino acid serves as a versatile intermediate in the preparation of advanced therapeutic candidates. It is frequently utilized in the synthesis of squaric acid derivatives which act as VLA-4 integrin antagonists, playing a significant role in immunological research. Additionally, it is employed in the construction of analogs related to kahalalide F, supporting investigations into bioactive marine natural products.
The nitro group modification offers distinct advantages for tuning the physicochemical properties of the final peptide structure. Researchers value this compound for its compatibility with standard Fmoc deprotection conditions using piperidine, ensuring smooth integration into automated synthesis workflows.
Quality Assurance and Storage
To preserve the integrity of the chemical structure, proper storage conditions are paramount. We recommend maintaining the product in a cool, dry environment at temperatures between 2-8°C. Containers should remain tightly closed to prevent moisture absorption, which could affect coupling efficiency during synthesis.
- Packaged in 25 kg drums or customized according to client requirements.
- Accompanied by a comprehensive Certificate of Analysis (COA) for every shipment.
- Manufactured under strict quality management systems to ensure batch-to-batch reproducibility.
Our commitment to quality extends beyond mere compliance. We provide global shipping solutions and technical support to ensure your research projects proceed without interruption. For bulk pricing and detailed manufacturing process information, please contact our sales team directly.



