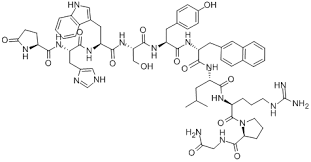
Nafarelin Acetate
- CAS No.76932-56-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Nafarelin Acetate API for pharmaceutical formulation. GMP compliant manufacturing with strict quality control and global supply capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
Nafarelin Acetate is a potent synthetic decapeptide analog of gonadotropin-releasing hormone (GnRH). As a critical active pharmaceutical ingredient (API), it functions as a superagonist, demonstrating significantly higher potency than the native hormone. Our manufacturing process ensures exceptional purity and structural integrity, making it suitable for downstream pharmaceutical formulation and clinical research applications. This peptide is extensively utilized in the development of therapeutic solutions for hormone-dependent conditions.
We specialize in the large-scale synthesis and supply of complex peptide APIs. Our facility adheres to rigorous quality management systems, ensuring that every batch of Nafarelin Acetate meets international pharmacopoeia standards. By leveraging advanced solid-phase peptide synthesis techniques, we maintain consistent quality and supply stability for our global partners.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | Nafarelin Acetate |
| CAS Number | 76932-56-4 |
| Molecular Formula | C66H83N17O13 |
| Molecular Weight | 1322.471 g/mol |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥98.0% |
| Density | 1.5±0.1 g/cm3 |
| Refractive Index | 1.711 |
| Packaging | 25 kg/drum or customized |
Industrial Applications
As a high-performance GnRH agonist, this compound serves as a key building block in the pharmaceutical industry. It is primarily indicated for the formulation of medications targeting central precocious puberty and endometriosis. Our clients utilize this API to develop controlled-release formulations and nasal spray solutions that require precise dosing and high bioavailability.
- Pharmaceutical formulation for hormone regulation therapies
- Research and development of novel peptide delivery systems
- Production of generic equivalents for established therapeutic brands
- Clinical trial material supply for regulatory submissions
Quality Assurance and Storage
Quality is paramount in peptide manufacturing. Each production batch undergoes comprehensive analytical testing, including HPLC, mass spectrometry, and microbial limits testing. We provide a complete Certificate of Analysis (COA) with every shipment to verify compliance with specified parameters. Our supply chain is optimized for temperature-controlled logistics to preserve peptide stability during transit.
For optimal stability, store the material in a cool, ventilated place away from direct sunlight and moisture. Proper storage conditions ensure the longevity and efficacy of the product throughout its shelf life. We offer flexible packaging options to meet specific manufacturing requirements, ensuring safe handling and minimal waste during production processes.



