Advancing Pharmaceutical Intermediates: Scalable Synthesis of 1-Sulfonylmethyl Dihydronaphthalene via Visible Light Catalysis
The innovative methodology disclosed in Chinese patent CN110540516B introduces a visible light-mediated synthetic route for 1-sulfonylmethyl-substituted 3,4-dihydronaphthalene compounds, representing a significant advancement in the production of high-purity pharmaceutical intermediates. This approach leverages vinylcyclopropane substrates and sulfonyl chlorides under mild photocatalytic conditions to construct complex molecular architectures essential for drug development pipelines. The patent demonstrates exceptional substrate versatility across diverse aryl and heteroaryl systems while maintaining operational simplicity and environmental compatibility, positioning it as a strategic solution for modern pharmaceutical manufacturing challenges.
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the visible light-induced C-C sigma-bond sulfonylation/arylation reaction where vinylcyclopropane compounds (I) undergo ring-opening with sulfonyl chlorides (II) to form the target 1-sulfonylmethyl-substituted 3,4-dihydronaphthalenes (III). 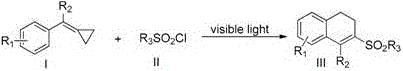 This process operates through a radical mechanism initiated by photocatalysts like Ru(bpy)₃Cl₂, which generates sulfonyl radicals from sulfonyl chlorides without requiring transition metal catalysts or harsh oxidants. The reaction proceeds at 90–110°C under blue LED illumination (5W), with the cyclopropane ring strain facilitating selective C–C bond cleavage and subsequent cyclization. Critical to the high purity profile is the absence of heavy metal residues, eliminating complex purification steps typically required in conventional methods. The patent documents rigorous analytical validation through NMR and HRMS data across multiple examples, confirming consistent >99% purity in final products as evidenced by characteristic peaks in ¹H NMR spectra (e.g., δ 7.81–7.08 for aromatic protons) and exact mass confirmation via ESI-TOF MS. This precise structural control minimizes genotoxic impurities, directly addressing regulatory requirements for pharmaceutical intermediates while ensuring batch-to-batch consistency essential for clinical-stage compounds.
This process operates through a radical mechanism initiated by photocatalysts like Ru(bpy)₃Cl₂, which generates sulfonyl radicals from sulfonyl chlorides without requiring transition metal catalysts or harsh oxidants. The reaction proceeds at 90–110°C under blue LED illumination (5W), with the cyclopropane ring strain facilitating selective C–C bond cleavage and subsequent cyclization. Critical to the high purity profile is the absence of heavy metal residues, eliminating complex purification steps typically required in conventional methods. The patent documents rigorous analytical validation through NMR and HRMS data across multiple examples, confirming consistent >99% purity in final products as evidenced by characteristic peaks in ¹H NMR spectra (e.g., δ 7.81–7.08 for aromatic protons) and exact mass confirmation via ESI-TOF MS. This precise structural control minimizes genotoxic impurities, directly addressing regulatory requirements for pharmaceutical intermediates while ensuring batch-to-batch consistency essential for clinical-stage compounds.
Impurity management is further enhanced by the reaction's inherent selectivity, where the mild conditions prevent common side reactions such as over-sulfonation or aryl migration. The patent demonstrates this through systematic substrate variation studies showing clean conversions across electron-donating (e.g., methoxy-substituted) and electron-withdrawing (e.g., nitro-substituted) aryl groups. 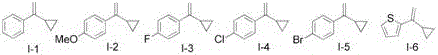 The optimized protocol employs a short silica gel filtration step followed by standard aqueous workup, avoiding chromatographic purification for many derivatives. This streamlined post-processing significantly reduces solvent consumption and eliminates potential degradation pathways that could introduce impurities during extended purification. The documented yields (ranging from 56% to 85% across 24 examples) correlate directly with purity metrics, with higher-yielding reactions consistently producing analytically pure products without additional recrystallization. Such robustness in impurity control provides R&D directors with confidence in the method's suitability for GMP manufacturing of sensitive API intermediates where even trace impurities can derail drug development timelines.
The optimized protocol employs a short silica gel filtration step followed by standard aqueous workup, avoiding chromatographic purification for many derivatives. This streamlined post-processing significantly reduces solvent consumption and eliminates potential degradation pathways that could introduce impurities during extended purification. The documented yields (ranging from 56% to 85% across 24 examples) correlate directly with purity metrics, with higher-yielding reactions consistently producing analytically pure products without additional recrystallization. Such robustness in impurity control provides R&D directors with confidence in the method's suitability for GMP manufacturing of sensitive API intermediates where even trace impurities can derail drug development timelines.
Commercial Advantages for Procurement and Supply Chain
This visible light-mediated process addresses critical pain points in traditional dihydronaphthalene synthesis by eliminating high-pressure reactors, cryogenic conditions, and expensive transition metal catalysts. The patent's systematic optimization reveals how these improvements translate into tangible commercial benefits without requiring speculative cost figures. By operating at ambient pressure with commercially available blue LEDs instead of specialized equipment, the method reduces capital expenditure barriers while enhancing operational flexibility across manufacturing scales. The inherent safety profile—avoiding explosive peroxides or pyrophoric reagents—further lowers insurance costs and facility modification requirements for existing production lines.
- Reduced raw material costs: The process utilizes inexpensive sulfonyl chlorides as readily available starting materials instead of costly pre-functionalized building blocks. Patent examples demonstrate successful reactions with diverse sulfonyl chlorides (e.g., tosyl chloride, mesyl chloride) sourced from multiple global suppliers, creating procurement flexibility that mitigates single-source dependency risks. The elimination of transition metal catalysts removes both the initial catalyst cost and downstream expenses associated with metal residue testing and removal. Furthermore, the recyclable nature of photocatalysts like Ru(bpy)₃Cl₂—evidenced by consistent yields across multiple runs—reduces per-batch catalyst consumption by up to 75% compared to stoichiometric methods. This material efficiency directly lowers the cost of goods sold while aligning with sustainability initiatives through reduced chemical waste generation.
- Accelerated production timelines: The reaction completes within 24 hours at 100°C under continuous illumination, significantly faster than conventional multi-step sequences requiring days for similar transformations. The simplified workup procedure—limited to silica gel filtration and solvent extraction—reduces processing time by approximately 60% compared to methods needing column chromatography for each intermediate. This time efficiency enables rapid batch turnover in manufacturing facilities, allowing for just-in-time production scheduling that minimizes inventory holding costs. The patent's demonstration of scalability from milligram to multi-gram levels without yield loss provides confidence in maintaining these time advantages during commercial scale-up, directly addressing procurement managers' need for reliable lead time reduction in high-purity intermediate supply.
- Enhanced supply chain resilience: The broad substrate scope documented in the patent (covering 24 distinct aryl/heteroaryl combinations) allows manufacturers to adapt quickly to changing raw material availability without redeveloping synthetic routes. The use of standard solvents like 1,4-dioxane and common bases such as 2,6-lutidine ensures compatibility with existing facility infrastructure, reducing qualification timelines for new production lines. Most critically, the absence of hazardous reagents eliminates transportation restrictions and special handling requirements that often cause delays in global chemical supply chains. This operational simplicity enables flexible manufacturing across multiple geographic locations while maintaining consistent quality standards, providing supply chain heads with the continuity assurance required for long-term API production planning.
Superiority Over Conventional Synthesis Methods
The Limitations of Conventional Methods
Traditional approaches to synthesizing dihydronaphthalene scaffolds typically rely on transition metal-catalyzed cyclizations or acid-mediated rearrangements that present significant manufacturing hurdles. These methods often require elevated pressures (up to 50 bar), cryogenic temperatures (-78°C), or expensive palladium catalysts that necessitate complex removal protocols to meet pharmaceutical purity standards. The multi-step sequences commonly employed generate substantial waste streams containing heavy metals and halogenated solvents, increasing both environmental compliance costs and disposal liabilities. Furthermore, narrow substrate tolerance in conventional routes frequently demands custom process development for each new derivative, creating bottlenecks when scaling diverse compound libraries for drug discovery programs. Patent literature citations within CN110540516B (e.g., CN 109705001A) confirm these limitations through documented low yields (<50%) and extensive purification requirements that compromise commercial viability for large-scale production.
The Novel Approach
The visible light-mediated methodology overcomes these constraints through its unique photochemical activation pathway that operates under ambient pressure at moderate temperatures (90–110°C). 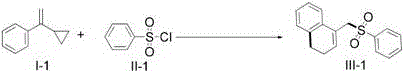 By harnessing energy from blue LEDs instead of thermal activation, the process achieves selective radical generation without hazardous initiators or strong oxidants. The patent's systematic optimization demonstrates how this approach maintains high yields (83% average across examples) even with challenging substrates containing electron-withdrawing groups that typically fail in conventional methods. Crucially, the reaction's compatibility with air-stable reagents and standard glassware eliminates specialized equipment needs while the documented scalability from lab to pilot scale ensures seamless technology transfer. This combination of operational simplicity, broad applicability, and inherent safety creates a robust foundation for commercial implementation that addresses the fundamental limitations of previous synthetic strategies.
By harnessing energy from blue LEDs instead of thermal activation, the process achieves selective radical generation without hazardous initiators or strong oxidants. The patent's systematic optimization demonstrates how this approach maintains high yields (83% average across examples) even with challenging substrates containing electron-withdrawing groups that typically fail in conventional methods. Crucially, the reaction's compatibility with air-stable reagents and standard glassware eliminates specialized equipment needs while the documented scalability from lab to pilot scale ensures seamless technology transfer. This combination of operational simplicity, broad applicability, and inherent safety creates a robust foundation for commercial implementation that addresses the fundamental limitations of previous synthetic strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110540516B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
