Advanced Visible Light Protocol for High-Purity 1-Sulfonylmethyl-3,4-dihydronaphthalene Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex polycyclic scaffolds efficiently. Patent CN110540516B introduces a groundbreaking synthetic approach for preparing 1-sulfonylmethyl-substituted 3,4-dihydronaphthalene compounds, a structural motif frequently encountered in bioactive molecules and advanced material precursors. This innovation leverages visible light mediation to drive the reaction between vinylcyclopropane compounds and sulfonyl chlorides, effectively bypassing the limitations of traditional thermal activation. By utilizing a photocatalytic system, the inventors have established a pathway that operates under significantly milder conditions while maintaining high target product yields. This development represents a significant leap forward for reliable pharmaceutical intermediates supplier networks, offering a greener and more versatile alternative to existing synthetic routes. The ability to functionalize dihydronaphthalene cores with sulfonyl groups opens new avenues for drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydronaphthalene derivatives has relied heavily on harsh thermal conditions or stoichiometric oxidants that generate substantial waste streams. Conventional strategies often involve multi-step sequences with poor atom economy, limiting their viability for cost reduction in pharmaceutical intermediates manufacturing. Prior art, such as the work by Jin-heng Li and Aiwen Lei, has explored visible light photocatalysis but often restricts the scope to specific substrates or requires expensive, non-recyclable catalysts. Furthermore, traditional sulfonylation methods using arylsulfonyl chlorides frequently suffer from low selectivity and the formation of difficult-to-remove impurities. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and increased production costs for high-purity fine chemicals. The reliance on strong acids or bases in older protocols also poses safety risks and equipment corrosion issues during commercial scale-up.
The Novel Approach
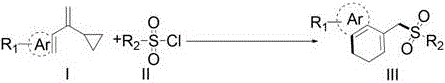
In stark contrast, the methodology disclosed in CN110540516B utilizes a visible light-mediated C-C sigma-bond sulfonylation and arylation cascade that fundamentally alters the reaction landscape. This novel approach enables the direct coupling of vinylcyclopropanes with sulfonyl chlorides under inert atmospheres, eliminating the need for aggressive oxidants. The reaction proceeds smoothly at temperatures between 90-110°C under LED illumination, demonstrating exceptional tolerance for various functional groups including halogens, nitro groups, and alkoxy substituents. By employing earth-abundant or recyclable photocatalysts like Ru(bpy)3Cl2, the process significantly reduces the environmental footprint associated with heavy metal contamination. This strategic shift not only enhances the purity profile of the final product but also simplifies the downstream purification processes, thereby streamlining the overall manufacturing workflow for complex organic molecules.
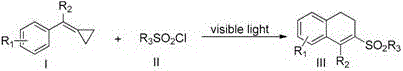
Mechanistic Insights into Visible Light Mediated C-C Sigma-Bond Sulfonylation
The core of this technological advancement lies in the intricate photocatalytic cycle that facilitates the homolytic cleavage of the sulfur-chlorine bond in sulfonyl chlorides. Upon irradiation with visible light, the photocatalyst enters an excited state, initiating a single-electron transfer (SET) process that generates highly reactive sulfonyl radicals. These radicals subsequently attack the electron-rich double bond of the vinylcyclopropane substrate, triggering a ring-opening event that is critical for forming the linear intermediate. This radical intermediate then undergoes an intramolecular cyclization onto the pendant aromatic ring, driven by the thermodynamic stability of the resulting dihydronaphthalene system. The precise control over radical generation ensures minimal side reactions, such as polymerization or over-oxidation, which are common pitfalls in free-radical chemistry. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters for specific substrate classes.
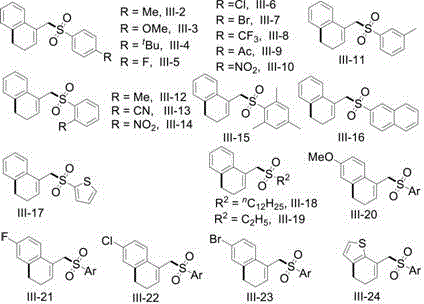
Furthermore, the reaction design incorporates a base-mediated deprotonation step that restores aromaticity and stabilizes the final sulfone product. The choice of base, such as 2,6-lutidine or pyridine, plays a pivotal role in scavenging the hydrochloric acid byproduct, preventing catalyst deactivation and substrate degradation. The structural versatility of the starting materials, defined by variable R1 and R2 groups, allows for the modular synthesis of a diverse library of derivatives. As illustrated in the patent data, substrates bearing electron-withdrawing or electron-donating groups on the aromatic rings participate efficiently, yielding products with consistent high quality. This mechanistic robustness ensures that the impurity profile remains manageable, facilitating easier regulatory approval for pharmaceutical applications where strict purity specifications are mandatory.
How to Synthesize 1-Sulfonylmethyl-3,4-dihydronaphthalene Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding atmosphere control and light source calibration to ensure reproducibility. The detailed standardized synthesis steps involve precise molar ratios of reactants and careful monitoring of reaction progress via TLC or GC-MS.
- Combine vinylcyclopropane compounds, sulfonyl chloride, photocatalyst (e.g., Ru(bpy)3Cl2), base, and organic solvent in a Schlenk tube under inert atmosphere.
- Illuminate the reaction mixture with visible light (e.g., 5W blue LED) while heating in an oil bath at 90-110°C for 12-36 hours.
- Filter the mixture, extract with ethyl acetate, dry over anhydrous Na2SO4, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible light protocol offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the substantial cost savings derived from the use of commercially available and inexpensive sulfonyl chlorides as starting materials. Unlike specialized organometallic reagents that require cold chain logistics and inert handling, sulfonyl chlorides are stable solids or liquids that can be sourced globally with minimal supply risk. Additionally, the elimination of stoichiometric oxidants and the use of catalytic amounts of photocatalysts drastically simplify the waste treatment process, reducing the operational expenditure associated with hazardous waste disposal. This streamlined process flow translates directly into improved margin potential for high-volume production runs.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts often required in traditional cross-coupling reactions, replacing them with more economical photocatalytic systems. By operating under milder thermal conditions, energy consumption for heating and cooling is optimized, leading to lower utility costs per kilogram of product. The high selectivity of the reaction minimizes the formation of byproducts, which reduces the solvent and silica gel consumption during the purification phase. Consequently, the overall cost of goods sold (COGS) is significantly lowered, enhancing competitiveness in the global fine chemicals market.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable reagents like vinylcyclopropanes and sulfonyl chlorides mitigates the risk of raw material shortages that often plague complex synthetic routes. The reaction's tolerance to a wide range of functional groups means that a single standardized protocol can be applied to synthesize multiple analogues, simplifying inventory management and production planning. This flexibility allows manufacturers to respond rapidly to changing market demands without the need for extensive process re-validation. Furthermore, the use of common organic solvents like 1,4-dioxane ensures that supply chains remain resilient against specialty solvent disruptions.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable using modern flow chemistry reactors or large-scale batch photoreactors, ensuring that laboratory success translates seamlessly to industrial production. The absence of heavy metal residues in the final product simplifies the regulatory compliance process, particularly for pharmaceutical intermediates destined for GMP facilities. By generating less hazardous waste and utilizing recyclable catalysts, the process aligns with increasingly stringent environmental regulations and corporate sustainability goals. This eco-friendly profile enhances the brand value of the supplier and facilitates long-term partnerships with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology.
Q: What are the key advantages of this visible light method over traditional thermal methods?
A: This method utilizes mild reaction conditions mediated by visible light, avoiding harsh thermal requirements and expanding substrate compatibility compared to conventional oxidative cross-coupling reactions.
Q: Which photocatalysts are most effective for this transformation?
A: The patent identifies Ru(bpy)3Cl2 as the preferred photocatalyst, though Ir(ppy)3 and Eosin Y derivatives also demonstrate efficacy depending on the specific substrate requirements.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the use of readily available sulfonyl chlorides and robust reaction conditions supports scalability, making it viable for commercial production of complex dihydronaphthalene scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Sulfonylmethyl-3,4-dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modern organic synthesis and have integrated these advanced capabilities into our CDMO service portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1-sulfonylmethyl-3,4-dihydronaphthalene meets the highest industry standards for pharmaceutical and fine chemical applications. Our commitment to quality and consistency makes us the preferred partner for companies seeking to secure their supply chain for critical intermediates.
We invite you to leverage our technical expertise to optimize your specific synthesis requirements and achieve your cost targets. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project volume. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of patent CN110540516B can drive value for your organization. Let us collaborate to bring your next generation of innovative molecules to market faster and more efficiently.
