Advanced Visible-Light Mediated Synthesis of 1-Sulfonylmethyl-3,4-dihydronaphthalene Intermediates
The pharmaceutical and fine chemical industries are constantly seeking efficient, sustainable, and cost-effective pathways to construct complex molecular scaffolds, particularly those containing sulfur motifs which are prevalent in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN110540516B, which discloses a novel preparation method for 1-sulfonylmethyl-substituted 3,4-dihydronaphthalene compounds. This technology leverages visible-light mediation to drive the reaction between vinylcyclopropane compounds and sulfonyl chlorides, offering a robust alternative to traditional thermal methods. For R&D directors and procurement specialists, this patent represents a pivotal shift towards greener chemistry that does not compromise on yield or substrate scope. The ability to synthesize these valuable dihydronaphthalene cores under mild conditions opens new avenues for the rapid assembly of diverse libraries of potential drug candidates and agrochemical intermediates, positioning this methodology as a critical asset for modern organic synthesis pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydronaphthalene frameworks and the introduction of sulfonyl groups have relied on methodologies that often suffer from significant operational drawbacks. Traditional approaches frequently necessitate the use of harsh reaction conditions, such as elevated temperatures and strong acidic or basic environments, which can lead to the decomposition of sensitive functional groups and limit the overall substrate tolerance. Furthermore, many existing protocols depend on expensive transition metal catalysts or specialized sulfur-containing reagents that are not only costly but also pose challenges regarding removal and residual metal contamination in the final active pharmaceutical ingredient (API). The reliance on stoichiometric oxidants in some oxidative cross-coupling reactions adds another layer of complexity, generating substantial waste and increasing the environmental footprint of the manufacturing process. These limitations collectively hinder the efficient scale-up and economic viability of producing sulfonyl-functionalized dihydronaphthalenes for commercial applications.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN110540516B introduces a streamlined, visible-light mediated strategy that fundamentally alters the reaction landscape. By utilizing readily available sulfonyl chlorides as radical precursors and vinylcyclopropanes as coupling partners, this method achieves the formation of C-C and C-S bonds simultaneously under remarkably mild conditions. 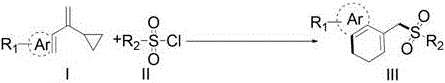 The use of visible light as the energy source eliminates the need for extreme thermal input, thereby preserving the integrity of delicate molecular structures. This approach not only simplifies the operational procedure but also significantly broadens the scope of compatible substrates, allowing for the incorporation of various electron-withdrawing and electron-donating groups without compromising reaction efficiency. The result is a versatile synthetic platform that delivers high yields of target products with exceptional purity profiles, making it an ideal candidate for industrial adoption.
The use of visible light as the energy source eliminates the need for extreme thermal input, thereby preserving the integrity of delicate molecular structures. This approach not only simplifies the operational procedure but also significantly broadens the scope of compatible substrates, allowing for the incorporation of various electron-withdrawing and electron-donating groups without compromising reaction efficiency. The result is a versatile synthetic platform that delivers high yields of target products with exceptional purity profiles, making it an ideal candidate for industrial adoption.
Mechanistic Insights into Visible-Light Mediated C-C Sigma-Bond Sulfonylation
The core of this innovation lies in the intricate photocatalytic cycle that drives the transformation. Upon irradiation with visible light, the photocatalyst—such as Ru(bpy)3Cl2 or organic dyes like Eosin Y—is excited to a higher energy state, enabling it to participate in single-electron transfer (SET) processes. This excitation facilitates the homolytic cleavage of the sulfur-chlorine bond in the sulfonyl chloride, generating a highly reactive sulfonyl radical species. This radical then adds to the double bond of the vinylcyclopropane moiety, triggering a cascade of events that includes the opening of the strained cyclopropane ring. The resulting carbon-centered radical undergoes an intramolecular cyclization with the pendant aromatic ring, ultimately leading to the formation of the dihydronaphthalene skeleton after rearomatization or hydrogen abstraction.  This mechanistic pathway is distinct from traditional ionic mechanisms and allows for the construction of complex cyclic systems that are otherwise difficult to access. The precise control over radical generation ensures that side reactions are minimized, leading to cleaner reaction profiles.
This mechanistic pathway is distinct from traditional ionic mechanisms and allows for the construction of complex cyclic systems that are otherwise difficult to access. The precise control over radical generation ensures that side reactions are minimized, leading to cleaner reaction profiles.
From an impurity control perspective, the mild nature of the photocatalytic conditions plays a crucial role in maintaining product quality. Unlike thermal methods that might promote polymerization or degradation of the vinylcyclopropane starting materials, the visible-light driven process operates at moderate temperatures (typically around 100°C in an oil bath, but driven by photon energy rather than thermal activation alone). This reduces the formation of thermal byproducts and oligomers. Furthermore, the selectivity of the radical addition to the alkene over other potential reactive sites ensures that the sulfonyl group is installed specifically at the desired position. The use of bases such as 2,6-lutidine helps to neutralize the hydrochloric acid byproduct generated during the reaction, preventing acid-catalyzed decomposition of the product or starting materials. This careful balancing of reaction parameters results in a crude product that is easier to purify, reducing the burden on downstream processing and ensuring a high-purity final API intermediate.
How to Synthesize 1-Sulfonylmethyl-3,4-dihydronaphthalene Efficiently
The practical implementation of this synthesis is designed for reproducibility and ease of handling in a standard laboratory or pilot plant setting. The protocol typically involves charging a Schlenk tube with the vinylcyclopropane substrate, the sulfonyl chloride coupling partner, a catalytic amount of photocatalyst, a suitable base, and an organic solvent such as 1,4-dioxane. The reaction vessel is then sealed and purged with an inert gas like argon to exclude oxygen, which could quench the radical intermediates. 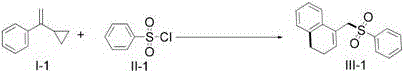 Once prepared, the reactor is placed in an oil bath and illuminated with a visible light source, such as a 5W blue LED lamp, while being stirred for a period ranging from 12 to 36 hours. The detailed standardized synthesis steps for this process are provided below.
Once prepared, the reactor is placed in an oil bath and illuminated with a visible light source, such as a 5W blue LED lamp, while being stirred for a period ranging from 12 to 36 hours. The detailed standardized synthesis steps for this process are provided below.
- Combine vinylcyclopropane compounds, sulfonyl chloride, photocatalyst (e.g., Ru(bpy)3Cl2), base, and organic solvent in a Schlenk tube under inert atmosphere.
- Place the reactor in an oil bath at 90-110°C and illuminate with visible light (e.g., 5W blue LED) while stirring for 12-36 hours.
- Filter the mixture, wash with ethyl acetate and brine, dry over anhydrous Na2SO4, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light mediated synthesis offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant reduction of raw material costs and processing complexity. By utilizing sulfonyl chlorides, which are commodity chemicals available in vast quantities and at low prices, the method avoids the need for expensive, custom-synthesized sulfur reagents. Additionally, the elimination of harsh thermal conditions and stoichiometric oxidants translates to lower energy consumption and reduced waste disposal costs, directly impacting the bottom line of manufacturing operations. The simplicity of the workup procedure, involving standard filtration and extraction techniques, further streamlines the production timeline, allowing for faster turnaround times from batch to batch.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by the use of earth-abundant or organic photocatalysts that can be employed at low loadings, minimizing the cost contribution of the catalyst to the final product. Unlike methods requiring precious metals like palladium or rhodium, the catalysts used here (such as Ruthenium complexes or Eosin Y) are relatively inexpensive and, in some cases, recoverable. The avoidance of cryogenic conditions or high-pressure equipment also means that existing standard glass-lined or stainless-steel reactors can be retrofitted with LED lighting arrays, avoiding massive capital expenditure on new infrastructure. This compatibility with existing assets ensures a rapid return on investment for manufacturers looking to upgrade their synthetic capabilities.
- Enhanced Supply Chain Reliability: The robustness of the reaction across a wide range of substrates ensures a stable supply of diverse intermediates. As demonstrated by the extensive substrate scope, the method tolerates various functional groups including halogens, nitriles, and ethers, which are common in pharmaceutical building blocks. This versatility means that supply chains are less vulnerable to disruptions caused by the unavailability of specific, highly specialized reagents. The reliance on common solvents like 1,4-dioxane or acetonitrile, which are widely stocked by chemical suppliers, further mitigates the risk of raw material shortages. Consequently, manufacturers can maintain consistent production schedules and meet the demanding delivery timelines of their global clients without interruption.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but advancements in LED technology and flow chemistry have largely overcome these barriers. The modular nature of LED arrays allows for linear scale-up, where increasing the surface area of illumination maintains reaction efficiency even at larger volumes. Moreover, the green chemistry credentials of this method align perfectly with increasingly stringent environmental regulations. The atom economy is favorable, and the reduction in hazardous waste generation simplifies compliance with environmental, health, and safety (EHS) standards. This makes the process not only commercially attractive but also socially responsible, enhancing the corporate image of manufacturers who adopt these sustainable practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light mediated synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the advantages of using visible light photocatalysis for dihydronaphthalene synthesis?
A: Visible light photocatalysis offers mild reaction conditions, avoiding harsh temperatures or pressures often required in thermal methods. It utilizes inexpensive and abundant sulfonyl chlorides as radical sources, significantly reducing raw material costs compared to specialized sulfur reagents.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method demonstrates wide substrate adaptability and high yields across various substituted vinylcyclopropanes and sulfonyl chlorides. The use of standard LED light sources and common organic solvents facilitates easy scale-up from laboratory to commercial manufacturing.
Q: What types of substituents are tolerated in this synthetic route?
A: The reaction tolerates a broad range of functional groups including alkyl, alkoxy, nitro, halogen, and cyano groups on both the aryl ring of the vinylcyclopropane and the sulfonyl chloride, allowing for diverse structural modifications essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Sulfonylmethyl-3,4-dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in the synthesis of complex pharmaceutical intermediates. Our team of expert chemists has extensively evaluated the methodology described in CN110540516B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are outfitted with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-sulfonylmethyl-3,4-dihydronaphthalene delivered meets the highest industry standards for quality and performance.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of novel analogs or large-scale production of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
