Advanced Photocatalytic Strategy for High-Purity 1-Sulfonylmethyl-3,4-Dihydronaphthalene Manufacturing
Introduction to Patent CN110540516B
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex cyclic scaffolds efficiently. Patent CN110540516B introduces a groundbreaking synthetic approach for preparing 1-sulfonylmethyl-substituted 3,4-dihydronaphthalene compounds, a structural motif frequently encountered in bioactive molecules and advanced material precursors. This innovation leverages visible light mediation to drive the reaction between vinylcyclopropane compounds and sulfonyl chlorides, effectively bypassing the need for harsh thermal conditions or stoichiometric oxidants often required in traditional radical chemistry. By utilizing a photocatalytic system, the inventors have achieved a transformation that not only expands the chemical space available to medicinal chemists but also aligns with green chemistry principles by operating under mild temperatures ranging from 90°C to 110°C. The significance of this technology lies in its ability to forge carbon-carbon and carbon-sulfur bonds simultaneously through a cascade process involving ring-opening and cyclization. 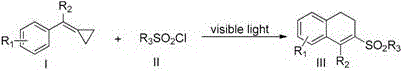 For procurement specialists and R&D directors alike, this represents a shift towards more sustainable and controllable manufacturing processes for high-value intermediates.
For procurement specialists and R&D directors alike, this represents a shift towards more sustainable and controllable manufacturing processes for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydronaphthalene derivatives has relied heavily on transition-metal catalyzed cyclizations or high-temperature thermal rearrangements that often suffer from poor atom economy and limited functional group tolerance. Traditional methods frequently require expensive palladium or rhodium catalysts, which introduce significant cost burdens and necessitate rigorous downstream purification to remove trace metal residues that are strictly regulated in pharmaceutical applications. Furthermore, classical radical sulfonylation strategies often depend on strong oxidants or UV light sources, which can lead to unselective side reactions, degradation of sensitive functional groups, and safety hazards associated with handling energetic reagents on a large scale. These conventional pathways typically exhibit narrow substrate scopes, failing to accommodate electron-deficient or sterically hindered substrates, thereby restricting the chemical diversity accessible to process chemists during lead optimization phases. The reliance on harsh conditions also complicates the engineering controls required for commercial production, increasing both capital expenditure and operational risks.
The Novel Approach
In stark contrast, the methodology disclosed in CN110540516B utilizes a visible-light-mediated strategy that operates under significantly milder conditions while delivering superior yields and broader substrate compatibility. By employing inexpensive and readily available sulfonyl chlorides as radical precursors, this novel route eliminates the need for pre-functionalized organometallic reagents, drastically simplifying the supply chain logistics for raw materials. The use of visible light, specifically blue or green LEDs, provides a clean energy source that activates the photocatalyst without generating the high-energy photons associated with UV degradation, thus preserving the integrity of complex molecular architectures. This approach enables the efficient construction of the dihydronaphthalene core through a tandem C-C sigma-bond cleavage and sulfonylation sequence, a transformation that is difficult to achieve with high selectivity using thermal methods alone. 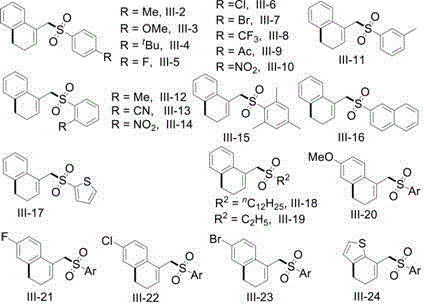 The versatility of this system is evidenced by its successful application to a wide array of substituted vinylcyclopropanes and sulfonyl chlorides, including those bearing halogens, nitro groups, and heterocycles, making it an invaluable tool for the rapid generation of diverse chemical libraries.
The versatility of this system is evidenced by its successful application to a wide array of substituted vinylcyclopropanes and sulfonyl chlorides, including those bearing halogens, nitro groups, and heterocycles, making it an invaluable tool for the rapid generation of diverse chemical libraries.
Mechanistic Insights into Visible Light Mediated C-C Sigma-Bond Sulfonylation
The core of this technological advancement lies in the intricate photocatalytic cycle that facilitates the generation of sulfonyl radicals and their subsequent addition to the vinylcyclopropane scaffold. Upon irradiation with visible light, the photocatalyst, typically a ruthenium or iridium complex such as Ru(bpy)3Cl2, enters an excited state capable of engaging in single-electron transfer (SET) processes with the sulfonyl chloride substrate. This interaction triggers the homolytic cleavage of the sulfur-chlorine bond, releasing a reactive sulfonyl radical and a chloride anion, while the photocatalyst returns to its ground state or undergoes a complementary redox cycle to maintain catalytic turnover. The generated sulfonyl radical then adds regioselectively to the terminal alkene of the vinylcyclopropane, initiating a cascade that involves the strain-driven ring-opening of the cyclopropane moiety. This ring-opening event generates a distal radical species that is perfectly positioned for an intramolecular cyclization onto the pendant aromatic ring, ultimately forming the stable dihydronaphthalene skeleton after rearomatization or hydrogen abstraction. 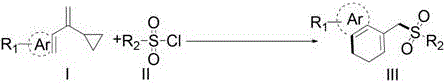 Understanding this mechanism is crucial for R&D teams as it highlights the precise control over radical intermediates, minimizing the formation of oligomeric byproducts that often plague free-radical reactions.
Understanding this mechanism is crucial for R&D teams as it highlights the precise control over radical intermediates, minimizing the formation of oligomeric byproducts that often plague free-radical reactions.
From an impurity control perspective, the mild nature of the visible light mediation plays a pivotal role in ensuring high product purity. Unlike thermal radical initiators that generate heat and can promote non-selective background reactions, the photochemical activation is spatially and temporally controlled by the light source, allowing for immediate cessation of radical generation when illumination stops. This controllability significantly reduces the incidence of over-sulfonylation or polymerization side reactions, leading to cleaner crude reaction profiles that simplify downstream purification. The choice of base, such as 2,6-lutidine, further aids in scavenging the hydrochloric acid byproduct formed during the reaction, preventing acid-catalyzed decomposition of the sensitive dihydronaphthalene product or the starting materials. Consequently, the final isolated products exhibit high chemical purity, reducing the burden on analytical quality control laboratories and ensuring that the material meets the stringent specifications required for pharmaceutical intermediate applications.
How to Synthesize 1-Sulfonylmethyl-3,4-Dihydronaphthalene Efficiently
The practical implementation of this synthesis is straightforward and relies on standard laboratory equipment, making it highly accessible for process development teams aiming to scale up production. The protocol involves charging a Schlenk tube with the vinylcyclopropane substrate, the sulfonyl chloride coupling partner, a catalytic amount of the photocatalyst, and a stoichiometric quantity of an organic base under an inert atmosphere to prevent oxygen quenching of the excited states. Following the addition of a polar aprotic solvent like 1,4-dioxane, the reaction mixture is subjected to visible light irradiation while being heated moderately to accelerate the kinetics without compromising selectivity. 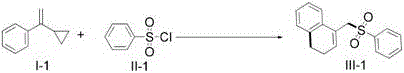 The detailed standardized synthesis steps for this high-efficiency route are outlined below.
The detailed standardized synthesis steps for this high-efficiency route are outlined below.
- Load vinylcyclopropane, sulfonyl chloride, photocatalyst (e.g., Ru(bpy)3Cl2), and base into a Schlenk tube under inert atmosphere.
- Add organic solvent such as 1,4-dioxane and seal the reactor.
- Illuminate with visible light (e.g., 5W blue LED) at 90-110°C for 12-36 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light mediated technology offers substantial strategic advantages regarding cost structure and operational reliability. The shift away from precious metal catalysts like palladium towards more abundant photocatalysts, combined with the use of commodity chemicals like sulfonyl chlorides, fundamentally alters the cost basis of producing these complex intermediates. Furthermore, the elimination of cryogenic conditions or high-pressure reactors simplifies the engineering requirements for manufacturing, allowing for production in standard glass-lined or stainless steel reactors equipped with LED arrays, which are increasingly common in modern multipurpose plants.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction workflow and the reduction in raw material costs. By utilizing sulfonyl chlorides, which are widely available and inexpensive compared to specialized organometallic reagents, the direct material cost is significantly lowered. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to a lower overall carbon footprint and utility cost per kilogram of product. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which translates to higher overall yields and reduced solvent usage during purification, further enhancing the cost efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on robust and commercially available starting materials ensures a stable supply chain that is less susceptible to geopolitical disruptions or market volatility. Vinylcyclopropanes and sulfonyl chlorides are produced by numerous global suppliers, providing procurement teams with multiple sourcing options to mitigate risk. The simplicity of the reaction setup also means that the technology can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without the need for specialized proprietary equipment, ensuring continuity of supply even in the face of unexpected production interruptions at a single facility.
- Scalability and Environmental Compliance: Scaling this photochemical process is facilitated by the modular nature of LED lighting systems, which can be arranged to provide uniform irradiation in larger reactors or flow chemistry setups. The absence of toxic heavy metal catalysts and strong oxidants simplifies waste treatment protocols, making it easier to comply with increasingly stringent environmental regulations regarding effluent discharge. The aqueous workup described in the patent, involving simple extraction and drying, generates waste streams that are easier to manage compared to those containing heavy metal residues, supporting the company's sustainability goals and reducing the costs associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, providing clarity for partners evaluating its potential for their specific projects. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation to ensure accuracy and relevance.
Q: What are the optimal reaction conditions for this sulfonylation?
A: The patent specifies using Ru(bpy)3Cl2 as the photocatalyst, 2,6-lutidine as the base, and 1,4-dioxane as the solvent at 100°C under 5W blue LED illumination for 24 hours.
Q: Does this method support heteroaryl substrates?
A: Yes, the method demonstrates wide substrate adaptability, successfully accommodating heteroaryl groups such as thienyl in the vinylcyclopropane component.
Q: Is the process scalable for industrial production?
A: The process utilizes mild conditions and standard workup procedures like filtration and extraction, making it highly suitable for commercial scale-up without requiring extreme pressure or temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Sulfonylmethyl-3,4-Dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light mediated synthesis in accelerating the development of next-generation pharmaceuticals and fine chemicals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-sulfonylmethyl-3,4-dihydronaphthalene delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can optimize your specific supply chain and reduce your time to market. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in photocatalytic processes can add tangible value to your project portfolio.
