Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Commercial Scale-Up
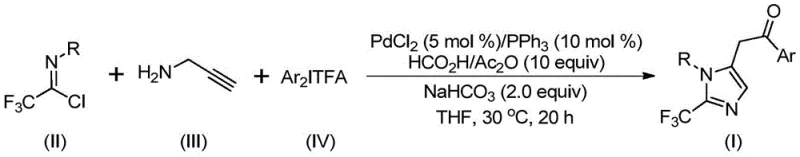
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which are pivotal scaffolds in modern drug design. Patent CN111423381A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical challenges in synthetic efficiency and safety. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing trifluoroacetimidoyl chloride, propargylamine, and diaryliodonium salts as key starting materials. The significance of this innovation lies in its ability to introduce the trifluoromethyl group—a moiety known to enhance metabolic stability, lipophilicity, and bioavailability—under remarkably mild conditions of 30°C. For R&D directors and procurement specialists, this represents a shift towards safer, more cost-effective manufacturing routes for high-value pharmaceutical intermediates, eliminating the need for hazardous diazo reagents often associated with traditional trifluoromethylation strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the use of highly reactive and potentially unstable synthons such as trifluorodiazoethane. These conventional approaches often necessitate stringent safety protocols, specialized equipment to handle explosive precursors, and harsh reaction conditions that can limit substrate compatibility. Furthermore, the direct reaction of trifluoromethyl synthons with suitable substrates frequently suffers from poor regioselectivity and limited functional group tolerance, leading to complex purification processes and reduced overall yields. The reliance on such dangerous reagents not only escalates the operational risk profile for manufacturing facilities but also significantly inflates the cost of goods sold due to the need for extensive safety infrastructure and waste management protocols, making large-scale production economically challenging for many organizations.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a palladium-catalyzed carbonylation strategy that operates under exceptionally mild conditions, typically at 30°C for 16 to 24 hours. By employing cheap and readily available starting materials like trifluoroacetimidoyl chloride and propargylamine, combined with diaryliodonium salts as oxidants, this process circumvents the hazards associated with diazo compounds. The reaction mechanism facilitates a cascade sequence that efficiently constructs the imidazole core while simultaneously installing the trifluoromethyl group with high precision. This approach not only simplifies the operational workflow but also broadens the utility of the method by accommodating a wide range of substituents, thereby enabling the synthesis of diverse libraries of 2-trifluoromethyl imidazoles essential for drug discovery and development programs.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated example of transition metal catalysis, initiating with a base-promoted intermolecular carbon-nitrogen bond formation to generate a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization followed by palladium-catalyzed aminopalladation of the alkyne moiety, yielding a vinyl-palladium intermediate. A critical step involves the isomerization to an alkyl-palladium species, which then undergoes carbonylation driven by carbon monoxide released in situ from the formic acid and acetic anhydride mixture. The resulting acyl-palladium intermediate is then subjected to oxidative addition by the diaryliodonium salt, forming a high-valent tetravalent palladium species. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole product and regenerates the active palladium catalyst, ensuring high turnover numbers and catalytic efficiency throughout the reaction duration.
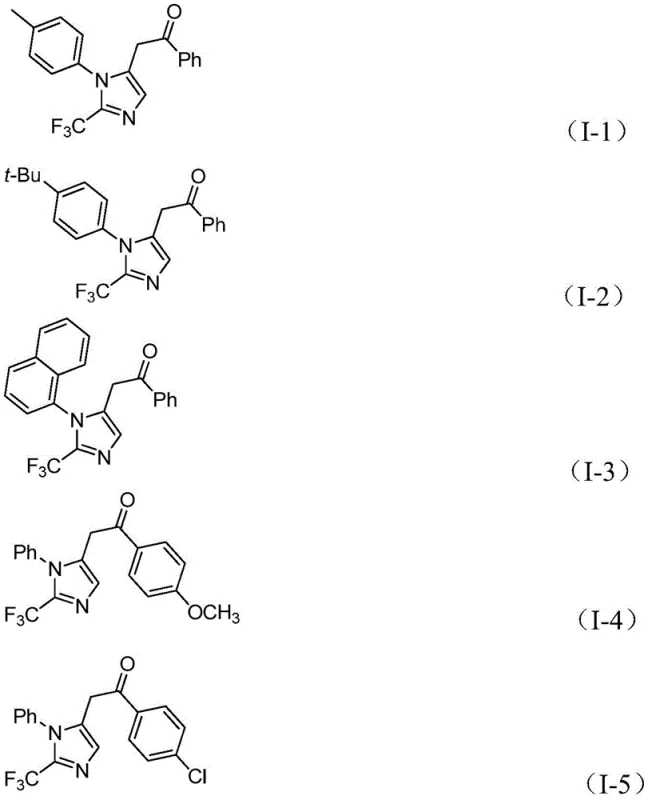
From an impurity control perspective, this mechanism offers distinct advantages due to the specificity of the palladium coordination and the mild reaction environment. The use of sodium bicarbonate as a base helps maintain a neutral to slightly basic pH, minimizing side reactions such as hydrolysis of the sensitive imidoyl chloride or polymerization of the alkyne. Furthermore, the in situ generation of carbon monoxide prevents the accumulation of toxic gas, enhancing process safety. The high functional group tolerance observed, allowing for substituents like methyl, tert-butyl, chloro, bromo, and nitro groups on the aryl rings, suggests that the catalytic cycle is robust against electronic variations. This robustness is crucial for maintaining high purity profiles in the final API intermediates, reducing the burden on downstream purification units and ensuring consistent quality for regulatory compliance.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The synthesis protocol outlined in the patent provides a standardized procedure that balances reaction efficiency with operational simplicity, making it highly attractive for process chemistry teams aiming to scale up production. The method involves a one-pot reaction where all components, including the palladium catalyst system, base, and reagents, are combined in an aprotic organic solvent such as tetrahydrofuran (THF). The detailed standard operating procedure ensures reproducibility and high conversion rates, with the patent explicitly noting that the reaction can be expanded to the gram level, providing a clear pathway for industrial application. For those interested in the specific stoichiometric ratios and workup procedures required to achieve yields as high as 97%, please refer to the standardized synthesis guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and the formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroacetimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic benefits regarding cost structure and supply continuity. The shift away from hazardous specialty reagents towards commodity chemicals like aniline derivatives and propargylamine drastically reduces raw material volatility and sourcing risks. Additionally, the mild reaction temperature of 30°C significantly lowers energy consumption compared to traditional high-temperature cyclization methods, contributing to a reduced carbon footprint and lower utility costs per kilogram of product. The simplicity of the post-processing, which involves filtration and standard column chromatography, minimizes the need for complex separation technologies, thereby streamlining the manufacturing timeline and reducing labor overheads associated with prolonged purification steps.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive catalysts such as palladium chloride and triphenylphosphine, which are used in relatively low loadings (5 mol% and 10 mol% respectively). By eliminating the need for expensive and dangerous trifluorodiazoethane, the direct material costs are significantly optimized. Furthermore, the high reaction efficiency and yields reported, reaching up to 97% in optimized examples, mean that less raw material is wasted, directly improving the atom economy and reducing the cost of goods sold. The avoidance of high-pressure equipment for carbon monoxide introduction, relying instead on liquid precursors, further reduces capital expenditure requirements for reactor infrastructure.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various substituted aromatic amines and diaryliodonium salts, are widely available from global chemical suppliers, ensuring a stable and resilient supply chain. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by environmental fluctuations or minor variations in reagent quality. This reliability is critical for maintaining continuous production schedules for key pharmaceutical intermediates, preventing stockouts that could delay downstream drug formulation. The ability to source precursors from multiple vendors reduces dependency on single-source suppliers, mitigating geopolitical and logistical risks inherent in the global chemical trade.
- Scalability and Environmental Compliance: The patent explicitly highlights the potential for scaling this method to industrial levels, supported by the use of common solvents like THF and straightforward workup procedures. The mild conditions reduce the generation of thermal waste and lower the risk of runaway reactions, aligning with modern green chemistry principles and stringent environmental regulations. The efficient use of reagents and the high selectivity of the catalytic system minimize the formation of by-products, simplifying waste treatment and disposal processes. This environmental compatibility not only ensures regulatory compliance but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios for complex heterocyclic compounds.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method avoids the use of hazardous trifluorodiazoethane and utilizes mild conditions (30°C) with readily available starting materials like trifluoroacetimidoyl chloride, offering superior safety and operational simplicity.
Q: What is the substrate scope for the aryl groups in this synthesis?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents on both the imidoyl chloride and the diaryliodonium salt, including electron-donating groups like methoxy and electron-withdrawing groups like nitro and halogens.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and potentially larger scales due to the use of cheap catalysts, simple post-processing, and mild reaction temperatures that reduce energy costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe management of palladium catalysts and the precise control of reaction parameters necessary to achieve stringent purity specifications. With our rigorous QC labs and commitment to quality assurance, we guarantee that every batch of 2-trifluoromethyl imidazole meets the highest international standards for drug substance manufacturing.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these critical building blocks for your innovative therapies.
