Scalable Electrochemical N-N Coupling for High-Purity Hydrazine Intermediates
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to construct nitrogen-nitrogen bonds, which are critical scaffolds in active pharmaceutical ingredients (APIs) and functional materials. Patent CN112126942B introduces a groundbreaking electrochemical methodology that achieves the N-N coupling of secondary aromatic amines to form tetra-substituted hydrazine compounds. This technology represents a significant paradigm shift from traditional stoichiometric oxidation to a catalytic-free, electron-driven process. By utilizing electricity as the primary reagent, this method avoids the generation of hazardous waste associated with chemical oxidants, aligning perfectly with modern green chemistry principles. For R&D directors and process chemists, this patent offers a robust alternative for synthesizing valuable hydrazine intermediates used in rocket propellants, antibacterial drugs, and anticancer agents, ensuring a cleaner impurity profile and simplified workup procedures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N-polysubstituted hydrazine compounds has relied heavily on transition metal catalysis, organometallic reagents, or the use of harsh chemical oxidants. These conventional routes often suffer from significant drawbacks, including the requirement for expensive noble metal catalysts such as palladium or copper, which necessitate rigorous and costly removal steps to meet pharmaceutical purity standards. Furthermore, the use of strong oxidants frequently leads to poor atom economy and the generation of substantial amounts of toxic by-products, creating environmental burdens and increasing waste disposal costs. Many existing methods also exhibit limited substrate scope, failing to efficiently couple sterically hindered or electronically diverse amines, which restricts their utility in complex molecule synthesis. Additionally, some protocols require stringent conditions such as nitrogen protection or the addition of strong base additives, complicating the operational procedure and increasing safety risks in large-scale manufacturing environments.
The Novel Approach
In stark contrast, the electrochemical method disclosed in CN112126942B utilizes electrons as clean reagents to drive the oxidative dehydrogenation coupling, theoretically releasing only hydrogen gas as a by-product. This approach operates under mild conditions in an undivided single-compartment cell, eliminating the need for external chemical oxidants or transition metal catalysts entirely. The process employs simple, commercially available starting materials like N-methylaniline analogues and inexpensive electrolytes such as sodium nitrite, making it highly economically viable. The reaction proceeds through a sequential anodic oxidation and cathodic reduction mechanism, enabling efficient self-coupling of the amine substrates with high atom utilization. This not only simplifies the reaction setup but also drastically reduces the environmental footprint, making it an ideal candidate for sustainable cost reduction in pharmaceutical intermediates manufacturing.

Mechanistic Insights into Electrochemical Oxidative Dehydrogenation
The core of this innovation lies in the precise control of electrode potentials to facilitate the formation of nitrogen-centered radicals without over-oxidation. In the anodic compartment, the secondary aromatic amine undergoes a single-electron transfer (SET) oxidation to generate a radical cation intermediate. This reactive species subsequently loses a proton to form a neutral nitrogen radical, which then dimerizes to form the N-N bond characteristic of the hydrazine structure. Simultaneously, the cathodic process likely involves the reduction of protons to hydrogen gas, maintaining charge balance within the undivided cell. The use of a graphite carbon rod anode and a platinum sheet cathode provides a stable surface for these electron transfers, ensuring consistent reaction kinetics. The presence of the electrolyte, specifically sodium nitrite in the optimized protocol, enhances the conductivity of the solution and may participate in mediating the electron transfer, thereby lowering the energy barrier for the oxidation step.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for producing high-purity tetra-substituted hydrazine compounds. Since no transition metals are introduced into the reaction system, the risk of heavy metal contamination, a critical failure point in API synthesis, is completely eradicated. This simplifies the downstream purification process, often requiring only standard extraction and column chromatography rather than specialized scavenging resins. Furthermore, the mild electrochemical conditions minimize side reactions such as over-oxidation to azo compounds or decomposition of sensitive functional groups on the aromatic rings. The patent data indicates that the method tolerates various substituents, including electron-donating and electron-withdrawing groups, suggesting a robust mechanism that can handle diverse electronic environments without compromising yield or selectivity.
How to Synthesize Tetra-Substituted Hydrazine Efficiently
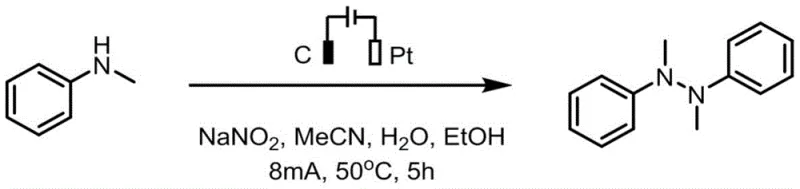
To implement this technology in a laboratory or pilot setting, the process follows a straightforward constant-current electrolysis protocol using readily available equipment. The reaction mixture typically consists of the amine substrate, a supporting electrolyte like sodium nitrite, and a mixed solvent system of acetonitrile, water, and ethanol to optimize solubility and conductivity. The simplicity of the setup, involving a standard undivided cell or even a three-necked flask equipped with electrodes, makes it accessible for rapid screening and optimization. Detailed operational parameters, including current density, temperature, and reaction time, have been rigorously optimized in the patent examples to ensure maximum conversion and yield. The following guide outlines the standardized steps derived from the preferred embodiments to assist process engineers in replicating this efficient synthesis.
- Prepare the electrolytic cell by adding N-methylaniline analogue, sodium nitrite electrolyte, and a mixed solvent of acetonitrile, water, and ethanol.
- Insert a graphite carbon rod anode and a platinum sheet cathode into the undivided cell and initiate constant current electrolysis at 8mA.
- Maintain the reaction at 50°C for 5 hours with stirring, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology presents compelling opportunities to optimize the sourcing and production of hydrazine derivatives. By shifting away from precious metal catalysts and hazardous oxidants, manufacturers can achieve substantial cost savings through reduced raw material expenses and simplified waste management protocols. The reliance on commodity chemicals like graphite, platinum (which is durable and reusable), and simple salts ensures a stable supply chain不受 geopolitical fluctuations affecting rare earth or specialty catalyst markets. Moreover, the operational simplicity and safety of the electrochemical cell design reduce the need for complex engineering controls, facilitating faster deployment and reducing lead time for high-purity hydrazine intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a significant cost driver from the bill of materials, while also negating the need for costly metal scavenging steps during purification. The use of electricity as the primary reagent is inherently more efficient than stoichiometric chemical oxidants, leading to lower overall reagent consumption. Additionally, the mild reaction conditions reduce energy consumption related to heating or cooling, contributing to a leaner manufacturing cost structure. The high atom economy of the reaction means less raw material is wasted as by-products, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: The starting materials, such as N-methylaniline and sodium nitrite, are bulk commodities with established global supply chains, ensuring consistent availability and price stability. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this electrochemical process are easily sourced from multiple suppliers. The robustness of the method across different substrate types allows for flexibility in production planning, enabling manufacturers to switch between different hydrazine products without major retooling. This flexibility strengthens the supply chain against disruptions and supports just-in-time manufacturing strategies for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The patent explicitly mentions the adaptability of this method to microchannel reaction devices, which are ideal for continuous flow manufacturing and rapid commercial scale-up of complex hydrazine compounds. Continuous flow electrochemistry offers superior heat and mass transfer compared to batch processes, allowing for safer handling of reactive intermediates at larger scales. The green nature of the process, with minimal waste generation and no heavy metal discharge, simplifies regulatory compliance and environmental permitting. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand value of the final product in markets increasingly driven by ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical N-N coupling technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed coupling?
A: This method eliminates the need for expensive transition metal catalysts and strong chemical oxidants, significantly reducing downstream purification costs and heavy metal contamination risks while improving atom economy.
Q: Can this process be scaled for industrial production of hydrazine intermediates?
A: Yes, the patent highlights the use of simple undivided cells and mentions the potential for microchannel reaction devices, indicating strong feasibility for commercial scale-up and continuous flow manufacturing.
Q: What types of substrates are compatible with this N-N coupling protocol?
A: The method demonstrates wide substrate applicability, successfully coupling symmetric and asymmetric diaryl amines, heteroaryl substrates, and monoaryl substrates like N-methylaniline and diphenylamine with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Substituted Hydrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of critical pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle electrochemical processes with precision, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-quality hydrazine derivatives that meet the exacting standards of the global pharmaceutical industry, leveraging our technical expertise to optimize yield and minimize impurities.
We invite you to collaborate with us to explore how this green electrochemical technology can enhance your supply chain efficiency and product quality. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to this metal-free protocol. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your target molecules. Let us help you navigate the future of sustainable chemical manufacturing with confidence and reliability.
