Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113045503B introduces a transformative approach for the preparation of 2-trifluoromethyl substituted quinazolinone compounds, a structural scaffold prevalent in numerous bioactive agents ranging from antifungal to anticancer therapeutics. This innovation addresses the longstanding challenges associated with introducing trifluoromethyl groups into heterocyclic systems, traditionally a process plagued by expensive reagents and苛刻 reaction conditions. By leveraging a transition metal palladium-catalyzed carbonylation cascade, this technology enables the efficient assembly of these valuable intermediates from cheap and readily available starting materials, specifically trifluoroethylimidoyl chloride and various amines.
The significance of this technological breakthrough extends beyond mere academic interest, offering tangible benefits for the commercial production of high-purity pharmaceutical intermediates. The quinazolinone core is a privileged structure found in marketed drugs such as Methaqualone and Afloqualone, as well as investigational candidates targeting diverse pathological pathways. 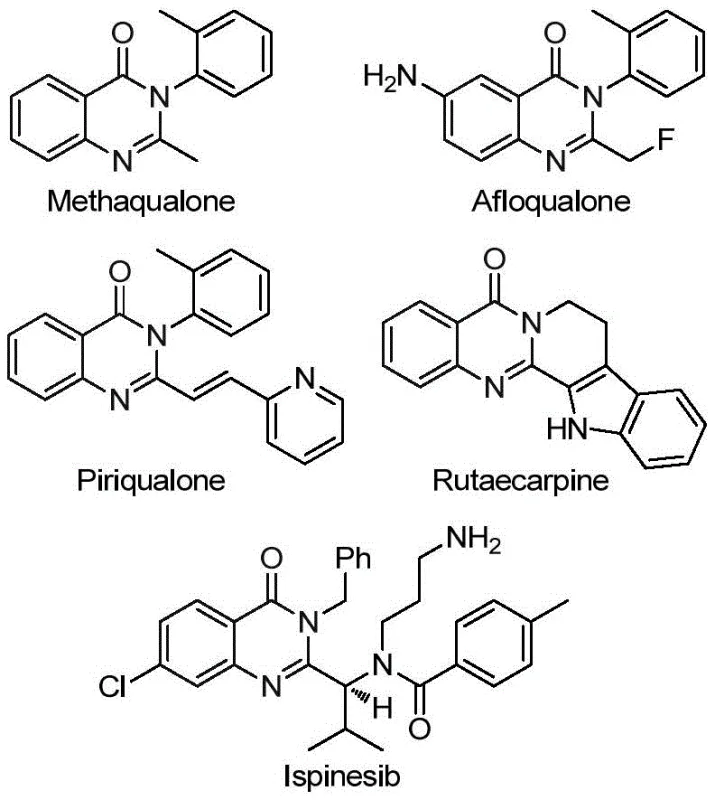 The ability to functionalize this core with a trifluoromethyl group at the 2-position significantly modulates the electronic and lipophilic properties of the parent molecule, often resulting in improved pharmacokinetic profiles. Consequently, reliable suppliers of these specialized intermediates are critical for accelerating drug discovery pipelines and ensuring the continuity of supply for essential medications.
The ability to functionalize this core with a trifluoromethyl group at the 2-position significantly modulates the electronic and lipophilic properties of the parent molecule, often resulting in improved pharmacokinetic profiles. Consequently, reliable suppliers of these specialized intermediates are critical for accelerating drug discovery pipelines and ensuring the continuity of supply for essential medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl quinazolinones has been hindered by significant synthetic bottlenecks that impede large-scale manufacturing. Conventional strategies often rely on the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, reactions that frequently necessitate harsh thermal conditions and generate substantial amounts of acidic waste. Alternative routes involving isatoic anhydride or T3P-promoted cascade reactions suffer from narrow substrate scopes and the requirement for pre-activated substrates, which drastically increases the step count and overall production costs. Furthermore, many existing protocols exhibit poor functional group tolerance, limiting their utility in the late-stage functionalization of complex drug candidates where sensitive moieties must be preserved.
The Novel Approach
In stark contrast, the methodology disclosed in CN113045503B utilizes a palladium-catalyzed carbonylation strategy that circumvents these traditional limitations through a streamlined one-pot process. By employing trifluoroethylimidoyl chloride as a stable electrophile and TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, the reaction proceeds under relatively mild conditions at 110°C in 1,4-dioxane. This approach not only eliminates the safety hazards associated with handling gaseous carbon monoxide but also ensures high reaction efficiency with excellent compatibility for diverse functional groups including halogens, alkyls, and aryls. The result is a versatile platform capable of generating a wide array of substituted quinazolinones with high purity, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps initiated by the interaction between the palladium catalyst and the organic substrates. Initially, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the trifluoroethylimidoyl chloride and the amine to form a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, generating a reactive divalent palladium intermediate. Under the heating conditions employed, TFBen decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form a crucial acyl palladium species.
Following the CO insertion, the presence of base facilitates the formation of a palladium-nitrogen bond, leading to the construction of a seven-membered ring palladium intermediate. The catalytic cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium species for further turnover. 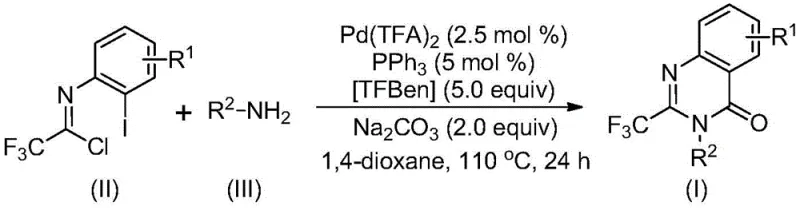 This intricate mechanism ensures high atom economy and minimizes the formation of side products, thereby simplifying the downstream purification process. For R&D directors, understanding this mechanism is vital for optimizing reaction parameters and troubleshooting potential issues during the commercial scale-up of complex pharmaceutical intermediates.
This intricate mechanism ensures high atom economy and minimizes the formation of side products, thereby simplifying the downstream purification process. For R&D directors, understanding this mechanism is vital for optimizing reaction parameters and troubleshooting potential issues during the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical implementation of this synthesis requires precise control over reaction stoichiometry and thermal conditions to maximize yield and minimize impurity profiles. The standard protocol involves charging a reactor with palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, the specific trifluoroethylimidoyl chloride, and the desired amine in an aprotic solvent. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated across multiple examples, are outlined below to guide process chemists in replicating this high-efficiency route.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and maintain stirring for 16 to 30 hours to ensure complete conversion via the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers distinct strategic advantages that directly impact the bottom line and operational resilience. The reliance on commercially available and inexpensive starting materials, such as various amines and trifluoroethylimidoyl chlorides, mitigates the risk of raw material shortages and price volatility often associated with exotic reagents. Furthermore, the use of a solid CO source instead of high-pressure gas cylinders significantly reduces infrastructure costs and safety compliance burdens, facilitating easier adoption in standard multipurpose manufacturing facilities without specialized gas handling equipment.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activation steps and the use of catalytic amounts of palladium significantly lower the direct material costs per kilogram of product. By avoiding harsh reagents that require specialized containment and neutralization, the process also reduces waste disposal expenses and extends the lifespan of reactor vessels, contributing to substantial long-term operational savings.
- Enhanced Supply Chain Reliability: The broad substrate scope demonstrated in the patent implies that the supply chain is not dependent on a single, hard-to-source precursor. The ability to utilize a wide range of amines allows for flexible sourcing strategies, ensuring that production schedules remain uninterrupted even if specific raw material vendors face temporary disruptions, thereby securing the continuity of supply for critical drug intermediates.
- Scalability and Environmental Compliance: The reaction has been proven effective on a gram scale with potential for expansion to multi-kilogram batches, indicating strong scalability for commercial production. Additionally, the generation of fewer hazardous byproducts compared to traditional anhydride-based methods aligns with increasingly stringent environmental regulations, reducing the ecological footprint of the manufacturing process and simplifying regulatory approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for your specific project needs.
Q: What are the key advantages of this Pd-catalyzed method over traditional cyclization routes?
A: Unlike conventional methods requiring harsh conditions or unstable reagents like trifluoroacetic anhydride, this protocol utilizes stable trifluoroethylimidoyl chloride and solid CO sources (TFBen), offering milder conditions, broader substrate tolerance, and higher yields suitable for industrial scale-up.
Q: Can this synthesis method be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the high-yield total synthesis of Rutaecarpine, achieving an overall yield of 77% through a three-step sequence involving cyclization, acid treatment, and base hydrolysis.
Q: What represents the primary cost driver in this synthetic route?
A: The primary cost drivers are the palladium catalyst and the specialized imidoyl chloride starting material; however, the use of commercially available amines and the elimination of gaseous carbon monoxide handling significantly reduce operational expenditures and safety compliance costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating the delivery of life-saving medicines to the market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest global standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce your overall time to market.
