Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Triazole Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN110467579B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as critical cores in numerous bioactive molecules ranging from antifungal agents to kinase inhibitors. The introduction of a trifluoromethyl group into these structures is particularly valuable, as it significantly enhances metabolic stability, lipophilicity, and bioavailability, properties that are essential for modern drug design. This patent presents a transformative approach that leverages inexpensive starting materials and a non-metallic iodine promotion system to achieve high-yielding cyclization.
The relevance of this chemical architecture cannot be overstated when observing current therapeutic landscapes. As illustrated in the structural diversity of known bioactive molecules, the 1,2,4-triazole motif is a privileged structure found in drugs such as deferasirox and various CYP enzyme inhibitors. 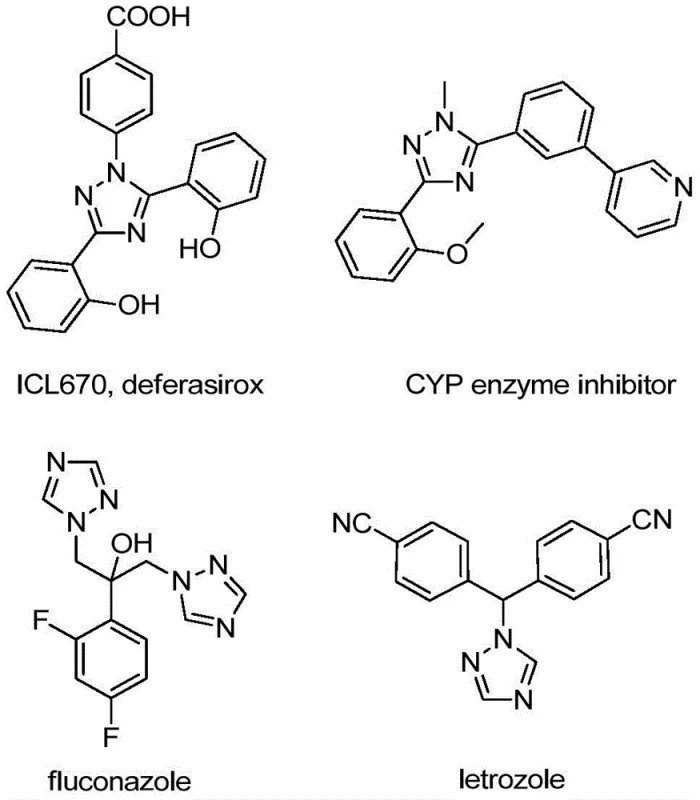 The ability to access these scaffolds with a trifluoromethyl substitution pattern opens new avenues for lead optimization and process chemistry. For R&D directors and procurement managers alike, understanding the nuances of this new synthetic methodology is crucial for securing a reliable supply chain of high-purity pharmaceutical intermediates. The method described offers a distinct advantage over traditional routes by simplifying the operational complexity while maintaining rigorous quality standards required for API production.
The ability to access these scaffolds with a trifluoromethyl substitution pattern opens new avenues for lead optimization and process chemistry. For R&D directors and procurement managers alike, understanding the nuances of this new synthetic methodology is crucial for securing a reliable supply chain of high-purity pharmaceutical intermediates. The method described offers a distinct advantage over traditional routes by simplifying the operational complexity while maintaining rigorous quality standards required for API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen-containing heterocycles has been fraught with significant technical and economic challenges. Traditional literature reports generally categorize existing methods into two main streams, both of which possess inherent drawbacks for large-scale manufacturing. The first approach involves the trifluoromethylation of pre-synthesized nitrogen-containing heterocycles. This pathway often necessitates the use of specialized and costly trifluoromethylating reagents, which can be hazardous to handle and difficult to source in bulk quantities. Furthermore, these reactions frequently require harsh conditions or sensitive catalytic systems that complicate the purification process and increase the overall cost of goods.
The second mainstream method utilizes synthons bearing a trifluoromethyl group, such as trifluorodiazoethane, reacting with coupling substrates. While effective in academic settings, reagents like trifluorodiazoethane are notoriously unstable and potentially explosive, posing severe safety risks in an industrial environment. Even the use of trifluoroethylimide acid halides, a safer alternative, has not been widely adopted due to limitations in reaction efficiency and substrate scope. These conventional pathways often demand strict anhydrous and anaerobic conditions, requiring specialized equipment and increasing the capital expenditure for manufacturing facilities. Consequently, there has been a persistent industry demand for a safer, more robust, and economically viable synthetic strategy.
The Novel Approach
The methodology outlined in patent CN110467579B represents a paradigm shift in how these valuable intermediates are constructed. By utilizing trifluoroethylimidoyl chloride and hydrazones as the primary building blocks, the process bypasses the need for dangerous diazo compounds or expensive late-stage trifluoromethylation reagents. The core innovation lies in the use of elemental iodine as a promoter in conjunction with sodium acetate. This combination facilitates a smooth cyclization under relatively mild thermal conditions, typically around 80°C to 100°C. The reaction scheme below highlights the elegance of this transformation, where simple mixing of reagents in a common organic solvent leads directly to the desired triazole core.
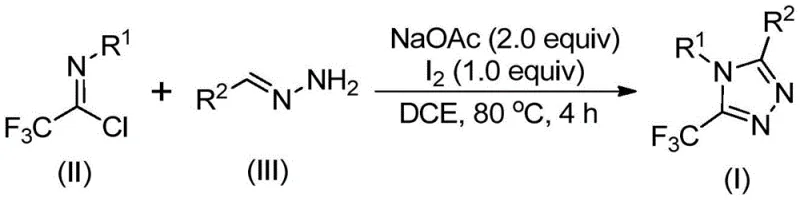 This novel approach drastically simplifies the operational workflow. Unlike previous methods, it does not require rigorous exclusion of moisture or oxygen, making it highly amenable to standard reactor setups found in most fine chemical plants. The use of inexpensive and commercially available starting materials, such as aromatic amines and aldehydes (used to prepare the hydrazones), ensures a stable and cost-effective supply chain. For procurement managers, this translates to reduced raw material volatility and lower input costs. Furthermore, the reaction demonstrates excellent functional group tolerance, allowing for the synthesis of diverse derivatives without the need for extensive protecting group strategies, thereby streamlining the overall synthetic sequence.
This novel approach drastically simplifies the operational workflow. Unlike previous methods, it does not require rigorous exclusion of moisture or oxygen, making it highly amenable to standard reactor setups found in most fine chemical plants. The use of inexpensive and commercially available starting materials, such as aromatic amines and aldehydes (used to prepare the hydrazones), ensures a stable and cost-effective supply chain. For procurement managers, this translates to reduced raw material volatility and lower input costs. Furthermore, the reaction demonstrates excellent functional group tolerance, allowing for the synthesis of diverse derivatives without the need for extensive protecting group strategies, thereby streamlining the overall synthetic sequence.
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the mechanistic underpinnings of this reaction is vital for R&D teams aiming to optimize the process for specific analogues. The transformation is believed to proceed through a multi-step cascade initiated by base-promoted intermolecular carbon-nitrogen bond formation. Initially, the hydrazone nucleophile attacks the electrophilic carbon of the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. This step is facilitated by sodium acetate, which acts as a mild base to scavenge the generated hydrochloric acid, driving the equilibrium forward without causing decomposition of sensitive functional groups. Following this initial condensation, the system undergoes an isomerization that aligns the reactive centers for the subsequent cyclization event.
The critical role of elemental iodine becomes apparent in the oxidative phase of the mechanism. It is hypothesized that iodine promotes an oxidative iodination of the intermediate, creating an electrophilic species susceptible to intramolecular nucleophilic attack. This triggers a cyclization followed by aromatization, ultimately yielding the stable 5-trifluoromethyl-1,2,4-triazole ring system. This mechanism avoids the use of transition metals like copper or palladium, which are common in cross-coupling reactions but leave behind toxic residues that are difficult to remove to ppm levels required for pharmaceuticals. By relying on iodine, a non-toxic and easily removable halogen, the impurity profile of the final product is significantly cleaner, reducing the burden on downstream purification units.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
Executing this synthesis requires careful attention to stoichiometry and reaction timing to maximize yield and purity. The patent data indicates that a molar ratio of trifluoroethylimidoyl chloride to hydrazone of approximately 1:2 is optimal, ensuring complete consumption of the limiting reagent. Sodium acetate is used in a 2.0 to 3.0 equivalent excess relative to iodine to maintain the necessary basicity throughout the reaction course. The choice of solvent is also critical; aprotic solvents such as 1,2-dichloroethane (DCE) have been shown to provide superior conversion rates compared to protic alternatives. Detailed standardized operating procedures for scaling this reaction from benchtop to pilot plant are essential for reproducibility.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE) within a reaction vessel.
- Heat the reaction mixture to a temperature range of 80°C to 100°C and maintain stirring for a duration of 2 to 4 hours to facilitate initial bond formation.
- Introduce elemental iodine into the system and continue heating for an additional 1 to 2 hours to promote oxidative cyclization and aromatization.
- Upon completion, perform standard post-treatment procedures including filtration and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the implications of this patented technology are profound. The shift away from precious metal catalysis and hazardous reagents directly impacts the cost structure of manufacturing these intermediates. By eliminating the need for expensive palladium or copper catalysts, the process removes a significant variable cost component. Moreover, the absence of heavy metals simplifies the purification workflow, potentially reducing the number of chromatography steps or crystallization cycles required to meet stringent regulatory specifications. This streamlined processing leads to substantial cost savings in terms of solvent usage, labor hours, and waste disposal fees.
- Cost Reduction in Manufacturing: The utilization of cheap, commodity-grade starting materials such as hydrazones and imidoyl chlorides creates a favorable economic baseline for production. Unlike specialized trifluoromethylating agents that command high market prices, these precursors are widely available from multiple global suppliers, fostering competitive pricing. Additionally, the avoidance of heavy metal catalysts means that manufacturers do not need to invest in expensive metal scavenging resins or complex recovery systems. The overall process efficiency, characterized by high conversion rates and minimal byproduct formation, ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Since the process does not require strict anhydrous or anaerobic environments, it is less susceptible to failures caused by equipment leaks or environmental fluctuations. The starting materials are stable and have long shelf lives, allowing for strategic stockpiling without degradation concerns. This stability reduces the risk of production delays caused by raw material spoilage or the need for just-in-time delivery of sensitive reagents. Consequently, manufacturers can offer more reliable lead times to their clients, ensuring uninterrupted production schedules for downstream drug formulations.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with green chemistry principles. The use of elemental iodine is preferable to toxic heavy metals, reducing the environmental footprint of the manufacturing process. The reaction can be easily scaled from gram quantities to multi-kilogram batches without significant re-engineering of the process parameters. The simplified workup procedure, involving basic filtration and standard chromatography, minimizes the generation of hazardous waste streams. This ease of scale-up and compliance with environmental regulations makes the technology highly attractive for long-term commercial partnerships and large-volume contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these details helps potential partners assess the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the practical realities of running this chemistry in a GMP-compliant environment.
Q: What are the primary advantages of this iodine-promoted synthesis method?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under mild conditions without strict anhydrous or anaerobic requirements, and utilizes cheap, readily available starting materials like hydrazones and trifluoroethylimidoyl chloride.
Q: What is the substrate scope for this triazole synthesis?
A: The process demonstrates broad functional group tolerance, successfully accommodating various substituted aryl groups (methyl, methoxy, bromo, trifluoromethyl) and heteroaryl groups (thiophene, pyridine, furan) at both the N1 and C5 positions of the triazole ring.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the protocol is designed for scalability. It avoids sensitive reagents and extreme conditions, allowing for easy expansion from gram-scale laboratory synthesis to multi-kilogram commercial production with simplified purification workflows.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN110467579B for the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex fluorinated heterocycles positions us as a preferred partner for global pharmaceutical companies seeking reliable sources of critical building blocks.
We invite you to collaborate with us to leverage this efficient synthesis route for your specific project needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. We encourage you to contact our technical procurement team to request specific COA data for related triazole analogues and to discuss route feasibility assessments for your target molecules. Together, we can accelerate your drug development timeline while maintaining the highest standards of quality and cost-efficiency.
