Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Global Supply Chains
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Global Supply Chains
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to bioactive scaffolds. Among these, 1,2,4-triazole derivatives stand out as critical structural motifs found in numerous pharmaceutical agents and functional materials, ranging from antifungal drugs to OLED ligands. A significant technological breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a novel preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This patent represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot oxidative cyclization strategy. By leveraging a metal-free catalytic system involving tetrabutylammonium iodide and tert-butyl hydroperoxide, this methodology addresses long-standing challenges in yield, operational simplicity, and environmental impact. For global procurement and R&D teams, understanding the nuances of this technology is essential for securing reliable supply chains of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale commercial application. Traditional protocols typically rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the desired heterocyclic framework. These legacy methods are characterized by severe reaction conditions that often require stringent control of temperature and pressure, alongside the use of stoichiometric amounts of activating agents. Furthermore, the cumulative yield of such multi-step processes is notoriously poor, often hovering around a mere 17% total yield. This low efficiency translates directly into excessive waste generation and inflated production costs, making these conventional routes economically unviable for the mass production of complex API intermediates required by the modern pharmaceutical industry.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN113307790B introduces a highly efficient, direct construction of the triazole ring using readily available 2-methylquinoline and trifluoroacetimidoyl hydrazide. This innovative approach bypasses the need for pre-functionalized carboxylic acids and eliminates the tedious multi-step sequences of the past. The core of this novelty lies in the use of a TBAI/TBHP catalytic system which promotes an oxidative cyclization directly from the methyl group of the quinoline. As illustrated in the general reaction scheme below, this transformation allows for the direct coupling of diverse substrates under relatively mild thermal conditions.
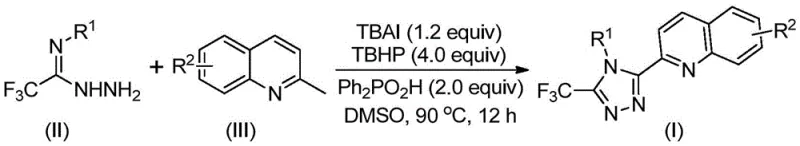
The versatility of this new route is further evidenced by its tolerance for a wide range of functional groups on both the quinoline and the hydrazide components. Substituents such as halogens, alkyl groups, and electron-donating methoxy groups are well-tolerated, enabling the synthesis of a diverse library of analogues without the need for protecting group strategies. This modularity is a critical advantage for medicinal chemists seeking to optimize structure-activity relationships (SAR) rapidly. Moreover, the reaction proceeds without the need for expensive transition metal catalysts, significantly reducing the burden of heavy metal removal during downstream processing and ensuring the final product meets stringent purity specifications for pharmaceutical applications.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The success of this synthetic route hinges on a sophisticated radical-mediated mechanism facilitated by the tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP) system. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate. Under the influence of the iodide catalyst and the peroxide oxidant, the methyl group is converted into a reactive 2-quinolinecarbaldehyde intermediate in situ. This transient aldehyde then undergoes a condensation reaction with the trifluoroacetimidoyl hydrazide to form a dehydrated hydrazone species. This step is crucial as it sets the stage for the subsequent ring closure.
Following the formation of the hydrazone intermediate, the system undergoes a series of oxidative transformations including oxidative iodination and intramolecular electrophilic substitution. The iodine species generated in the catalytic cycle acts as an electrophile, activating the hydrazone nitrogen for nucleophilic attack on the quinoline ring or facilitating the necessary bond formations to close the five-membered triazole ring. Finally, an aromatization step restores the aromaticity of the newly formed triazole system, driving the reaction to completion. The use of diphenylphosphoric acid as an additive further enhances the reaction efficiency, likely by stabilizing intermediates or facilitating proton transfer steps. This mechanistic pathway avoids the formation of stable by-products common in metal-catalyzed couplings, thereby simplifying the impurity profile and enhancing the overall robustness of the process for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
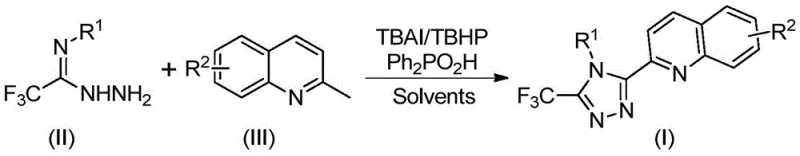
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal parameters to maximize yield and minimize side reactions. The protocol is designed to be operationally simple, utilizing common organic solvents like DMSO which effectively dissolve both the organic substrates and the ionic catalyst. The reaction is typically conducted by charging the reactor with the substrates, catalyst, oxidant, and additive, followed by heating to a specific temperature range. The specific experimental conditions and substrate variations demonstrated in the patent examples provide a robust template for replication.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphoric acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization and aromatization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical elegance. The shift away from precious metal catalysis and multi-step syntheses fundamentally alters the cost structure and risk profile of manufacturing these valuable intermediates. By simplifying the process flow and utilizing commodity chemicals, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing while simultaneously improving supply security.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper removes a major cost driver from the bill of materials. Furthermore, the avoidance of these metals negates the need for specialized and costly scavenging resins or complex purification steps required to meet residual metal limits in APIs. The use of cheap and commercially available starting materials like 2-methylquinoline and aqueous TBHP further drives down the raw material costs. The high yields reported, reaching up to 97% in optimized cases, mean that less raw material is wasted per kilogram of product, drastically improving the overall process economics and margin potential for high-volume production.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade reagents ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized fine chemicals or rare earth catalysts. The robustness of the reaction, which does not require strict anhydrous or anaerobic conditions, reduces the complexity of the manufacturing infrastructure needed. This ease of operation allows for production in a wider range of facilities, diversifying the supplier base and reducing the risk of disruption. The ability to source precursors from multiple vendors enhances negotiation leverage and ensures continuity of supply for critical drug development programs.
- Scalability and Environmental Compliance: The simplified workup procedure, involving filtration and standard column chromatography or crystallization, is inherently easier to scale than multi-step sequences with intermediate isolations. The metal-free nature of the process significantly reduces the environmental footprint by eliminating heavy metal waste streams, aligning with increasingly stringent global environmental regulations. This green chemistry profile facilitates faster regulatory approval and reduces the costs associated with waste treatment and disposal. The process is amenable to gram-scale and potentially multi-ton scale production, providing a clear pathway from R&D to commercial manufacturing without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their specific applications.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-carboxylic acid and five steps with harsh conditions yielding only 17%, this patent describes a one-pot, metal-free process using cheap 2-methylquinoline with yields up to 97%.
Q: Does this process require expensive transition metal catalysts?
A: No, the process utilizes a metal-free catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), eliminating the need for costly palladium or copper catalysts and simplifying purification.
Q: Is the reaction sensitive to moisture or oxygen?
A: The method is robust and does not require strict anhydrous or anaerobic conditions, as it utilizes an aqueous solution of TBHP and operates effectively in ambient atmospheric conditions, facilitating easier scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative cyclization technology described in Patent CN113307790B for the production of high-value heterocyclic intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthesis platform for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can accelerate your development timelines and reduce your overall cost of goods sold.
