Advanced Pd-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds for Commercial API Production
Introduction to Next-Generation Pyrrolinone Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and pharmaceutical agents. As highlighted in recent literature, compounds such as Althiomycin exhibit potent antibacterial properties, while Glimepiride functions as a critical hypoglycemic agent, and Isomalyngamide A demonstrates promising anticancer activity. The strategic construction of this heterocyclic core is therefore of paramount importance to the global pharmaceutical supply chain. A groundbreaking methodology detailed in patent CN112694430B introduces a highly efficient, palladium-catalyzed bis-carbonylation strategy that utilizes readily available propargyl amines and benzyl chlorides. This innovative approach circumvents traditional limitations by employing a solid carbon monoxide surrogate, thereby streamlining the synthetic pathway into a single, robust operation that is ideally suited for the scalable production of high-purity pharmaceutical intermediates.
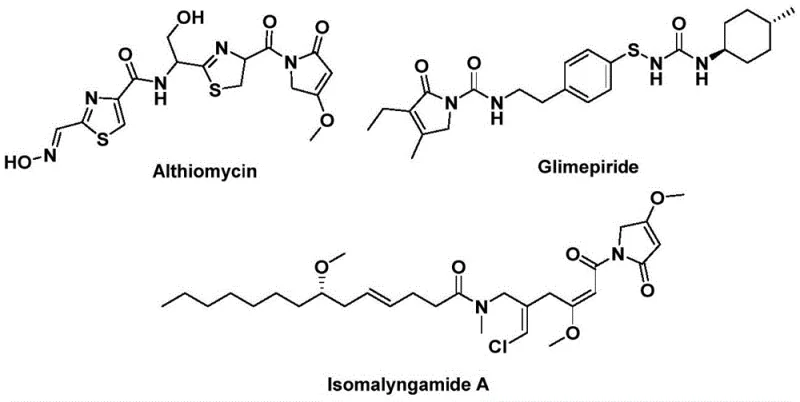
For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, the ability to access complex heterocycles through convergent, one-pot transformations is a significant value driver. The technology described herein not only expands the chemical space accessible for drug discovery but also offers a pragmatic solution for process intensification. By leveraging the unique reactivity of internal alkynes and benzylic halides under carbonylative conditions, this method delivers the target pyrrolinone derivatives with exceptional regioselectivity and yield. The versatility of this system allows for the rapid generation of diverse analog libraries, facilitating structure-activity relationship (SAR) studies while simultaneously providing a viable route for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 1,5-dihydro-2H-pyrrol-2-ones has relied heavily on the direct use of carbon monoxide gas. While effective in principle, the utilization of gaseous CO presents severe logistical and safety challenges, particularly in large-scale manufacturing environments. The requirement for high-pressure reactors, specialized gas handling infrastructure, and rigorous safety protocols to mitigate the risks of CO toxicity often results in prohibitive capital expenditures and extended lead times for high-purity intermediate production. Furthermore, traditional multi-step sequences often suffer from poor atom economy, requiring the installation and subsequent removal of protecting groups, which generates substantial chemical waste and increases the overall cost of goods sold (COGS). These inefficiencies create bottlenecks in the supply chain, making it difficult to respond agilely to market demands for key API precursors.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112694430B revolutionizes this landscape by introducing a gas-free carbonylation protocol. By utilizing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate, the process eliminates the hazards associated with high-pressure gas cylinders, allowing the reaction to proceed in standard laboratory glassware or simplified industrial reactors. This shift fundamentally alters the risk profile of the synthesis, enabling cost reduction in pharmaceutical intermediate manufacturing through simplified engineering controls. The reaction operates under relatively mild thermal conditions, typically between 100 °C and 120 °C, and achieves high conversion rates within 24 to 48 hours. This one-pot bis-carbonylation strategy merges bond formation and ring closure into a single operational step, drastically reducing solvent consumption and purification burdens compared to linear synthetic routes.
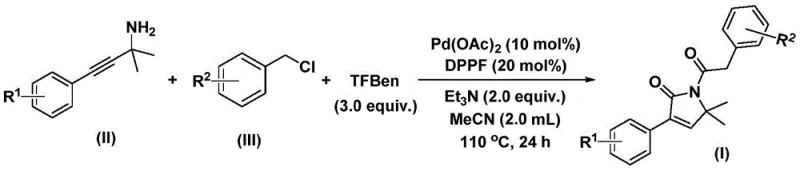
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
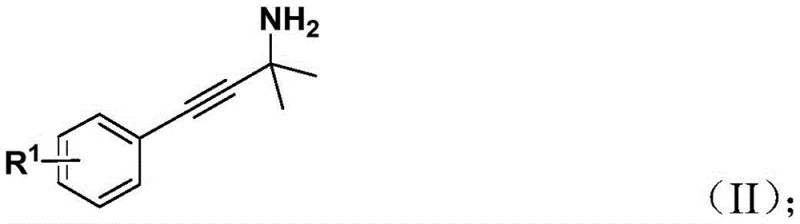
Understanding the catalytic cycle is essential for optimizing this process for commercial application. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium(II) intermediate. This step is facilitated by the bidentate phosphine ligand, 1,1'-bis(diphenylphosphino)ferrocene (DPPF), which stabilizes the metal center and promotes the subsequent migratory insertion. Following this activation, the first molecule of carbon monoxide, released in situ from the thermal decomposition of the phenol ester surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack or coordination with the propargyl amine substrate, initiating the cyclization event that constructs the five-membered nitrogen-containing ring.
The mechanistic elegance continues as a second carbon monoxide molecule inserts into the newly formed organopalladium species, expanding the coordination sphere and setting the stage for the final ring closure. This double carbonylation sequence is critical for establishing the 1,5-dihydro-2H-pyrrol-2-one core with the correct oxidation state and substitution pattern. Finally, reductive elimination releases the target product and regenerates the active palladium(0) catalyst, closing the loop. This intricate dance of bond formations is highly sensitive to the electronic nature of the substrates; however, the system demonstrates remarkable tolerance. Whether the aromatic rings bear electron-donating methoxy groups or electron-withdrawing trifluoromethyl moieties, the catalytic cycle proceeds efficiently, ensuring consistent quality and minimizing the formation of difficult-to-remove impurities that often plague transition metal-catalyzed processes.

How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
To implement this robust synthetic route, operators must adhere to precise stoichiometric ratios and thermal profiles to maximize yield and purity. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, solid CO source, and the two primary organic building blocks in an appropriate organic solvent such as acetonitrile. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined in the guide below to ensure reproducibility across different manufacturing sites.
- Combine palladium acetate, DPPF ligand, triethylamine, solid CO surrogate (TFBen), propargyl amine, and benzyl chloride in acetonitrile solvent within a Schlenk tube.
- Heat the reaction mixture to 110 °C and stir continuously for 24 hours under inert atmosphere to facilitate the bis-carbonylation cyclization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this patented technology offers transformative benefits that extend beyond mere chemical yield. The shift from gaseous reagents to solid surrogates fundamentally de-risks the supply chain, removing dependencies on specialized gas vendors and high-pressure logistics. This operational simplification translates directly into enhanced supply chain reliability, as production can be maintained even during disruptions in industrial gas supply networks. Furthermore, the use of commercially available starting materials like benzyl chlorides and propargyl amines ensures a stable upstream supply base, reducing the volatility of raw material costs and securing long-term availability for critical API intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements significantly lowers capital expenditure (CAPEX) for new production lines, while the one-pot nature of the reaction reduces operating expenditure (OPEX) by minimizing labor hours, solvent usage, and energy consumption associated with multiple isolation steps. By avoiding the need for expensive heavy metal scavengers often required to remove residual palladium from complex mixtures, the downstream processing costs are substantially optimized, leading to a more competitive price point for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on shelf-stable solid reagents rather than compressed gases enhances the resilience of the manufacturing process against external logistical shocks. The broad substrate scope means that a single production line can be rapidly repurposed to synthesize various analogs by simply swapping the starting benzyl chloride or amine, providing unparalleled flexibility to meet changing customer demands without extensive re-validation or equipment modification.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste compared to traditional multi-step syntheses, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities. The high atom economy and efficient conversion rates ensure that raw materials are utilized effectively, supporting sustainable manufacturing practices that are increasingly mandated by global regulatory bodies and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential applications for technical teams evaluating this route for integration into their existing portfolios.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous high-pressure CO gas, thereby significantly enhancing operational safety and simplifying reactor requirements.
Q: Does this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl substituents without compromising yield.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds in acetonitrile at elevated temperatures between 100 °C and 120 °C for a duration of 24 to 48 hours, utilizing a catalytic system of Pd(OAc)2 and DPPF with triethylamine as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
As the demand for complex heterocyclic scaffolds continues to rise in the development of next-generation therapeutics, having a manufacturing partner with deep technical expertise is crucial. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale industrial supply. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the 1,5-dihydro-2H-pyrrol-2-one intermediates you receive meet the highest industry standards.
We invite you to leverage our technical capabilities to optimize your supply chain and accelerate your drug development timelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative palladium-catalyzed process can deliver value to your organization.
