Advanced Palladium-Catalyzed Synthesis of Chiral Indole Intermediates for Commercial Antiviral Production
Advanced Palladium-Catalyzed Synthesis of Chiral Indole Intermediates for Commercial Antiviral Production
The pharmaceutical industry is constantly seeking efficient routes to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112961100B introduces a groundbreaking methodology for the synthesis of optically active indole compounds featuring cyano quaternary carbon chiral centers. This technology leverages a sophisticated palladium-catalyzed asymmetric cyano addition reaction of alkynyl amines, providing a direct and highly stereoselective pathway to structurally diverse indole derivatives. The significance of this innovation lies not only in its chemical elegance but also in its practical application, as the resulting compounds have demonstrated potent inhibitory effects against the Hepatitis B Virus (HBV). By targeting key viral replication markers such as HBV transcription and HBeAg secretion, these molecules represent a promising frontier in antiviral drug discovery. For research and development teams, this patent offers a robust platform for generating libraries of chiral indoles with high enantiomeric ratios, facilitating rapid structure-activity relationship studies.
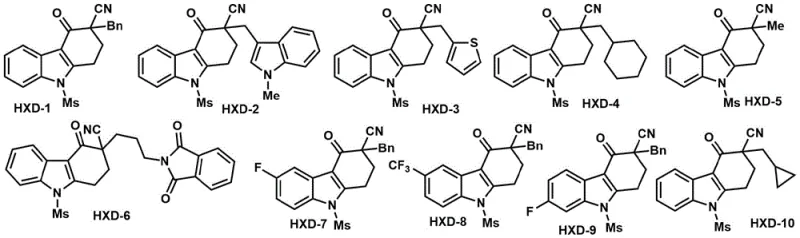
In the context of modern drug development, the ability to rapidly access enantiomerically pure intermediates is a decisive factor in project timelines. The disclosed method addresses the historical challenge of constructing all-carbon quaternary stereocenters within indole frameworks, a structural motif prevalent in many bioactive alkaloids and pharmaceutical agents. Unlike conventional approaches that often rely on tedious resolution techniques or stoichiometric chiral auxiliaries, this catalytic system achieves high levels of stereocontrol directly from prochiral substrates. The versatility of the reaction allows for the incorporation of various functional groups at multiple positions on the indole ring, enabling medicinal chemists to fine-tune pharmacokinetic properties without compromising synthetic efficiency. This level of molecular precision is essential for optimizing drug candidates that require specific spatial arrangements to interact effectively with biological targets, thereby accelerating the path from benchtop discovery to clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for accessing indole derivatives with quaternary carbon centers are frequently plagued by inefficiencies that hinder their scalability and economic viability. Conventional routes often involve multi-step sequences requiring harsh reaction conditions, expensive reagents, and extensive purification efforts to remove byproducts and impurities. A significant bottleneck in these legacy methods is the difficulty in establishing stereochemistry at the quaternary center, which typically necessitates the use of chiral pool starting materials that are limited in availability and structural diversity. Furthermore, classical cyclization protocols may suffer from poor regioselectivity, leading to mixtures of isomers that are difficult to separate, thereby reducing overall yield and increasing material costs. The reliance on stoichiometric amounts of chiral inducers not only drives up the cost of goods but also generates substantial chemical waste, posing environmental challenges that are increasingly scrutinized in green chemistry initiatives. These limitations collectively create a barrier to the rapid production of high-quality intermediates needed for advancing antiviral programs.
The Novel Approach
The innovative approach detailed in the patent overcomes these hurdles by employing a transition metal-catalyzed cascade reaction that constructs the indole core and the chiral quaternary center simultaneously. This palladium-catalyzed process utilizes readily available alkynyl amine precursors and malononitrile derivatives, which undergo an intramolecular cyclization driven by the formation of a stable palladium complex. The use of specialized chiral ligands, such as phosphino-oxazoline (Phox) derivatives, ensures exceptional enantioselectivity, consistently delivering products with high enantiomeric ratios suitable for pharmaceutical applications. This one-pot transformation significantly reduces the number of operational steps, minimizing solvent consumption and waste generation while maximizing atom economy. The reaction conditions are optimized to tolerate a wide range of substituents, allowing for the synthesis of diverse analogues without the need for protecting group manipulations. By streamlining the synthetic workflow, this novel approach offers a sustainable and cost-effective solution for producing complex chiral indoles.
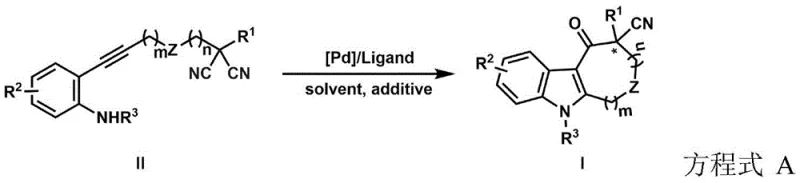
Mechanistic Insights into Palladium-Catalyzed Asymmetric Cyclization
The success of this synthetic methodology hinges on the intricate interplay between the palladium catalyst, the chiral ligand, and the substrate during the catalytic cycle. The reaction initiates with the coordination of the palladium species to the alkyne moiety of the substrate, activating it towards nucleophilic attack. Subsequent migratory insertion and reductive elimination steps facilitate the formation of the new carbon-carbon bonds that define the indole skeleton. The chiral environment provided by the ligand dictates the facial selectivity of the cyanide addition, ensuring that the quaternary carbon center is formed with the desired absolute configuration. Detailed optimization studies revealed that the nature of the ligand backbone and the steric bulk of the substituents play critical roles in achieving high enantioselectivity. For instance, ligands with specific steric profiles were found to suppress competing side reactions, such as polymerization or non-selective hydrolysis, thereby channeling the reaction pathway towards the desired cyclic product. Understanding these mechanistic nuances allows for the rational design of improved catalyst systems that can further enhance reaction rates and selectivity.
Impurity control is another critical aspect of this catalytic process, particularly when scaling up for commercial production. The reaction conditions are carefully tuned to minimize the formation of regioisomers and over-reacted byproducts that could complicate downstream purification. The use of specific additives, such as water or mild acids, helps to regulate the protonation state of intermediates, preventing the accumulation of unstable species that might degrade the final product. Additionally, the choice of solvent system influences the solubility of the catalyst and substrates, ensuring homogeneous reaction conditions that promote consistent kinetics throughout the batch. Rigorous monitoring of reaction parameters, including temperature and concentration, is essential to maintain the integrity of the chiral information transferred from the ligand to the product. By implementing strict process controls, manufacturers can ensure that the final indole intermediates meet the stringent purity specifications required for pharmaceutical use, reducing the risk of batch failures and ensuring supply chain reliability.
How to Synthesize Optically Active Indole Compounds Efficiently
The practical implementation of this synthesis involves a straightforward protocol that can be adapted for both laboratory-scale optimization and pilot-plant production. The process begins with the preparation of the active catalyst species by mixing a divalent palladium precursor with the chosen chiral ligand in an appropriate organic solvent. This mixture is stirred to allow for complete complexation before the addition of the alkynyl amine substrate and the cyanide source. The reaction is then heated to the optimal temperature, typically ranging from 80°C to 120°C, to drive the cyclization to completion. Monitoring the reaction progress via thin-layer chromatography or HPLC ensures that the conversion is maximized while preventing over-reaction. Upon completion, the reaction mixture is cooled and subjected to a standard workup procedure involving acid quenching and extraction to isolate the crude product. Final purification is achieved through column chromatography using standard silica gel media, yielding the target indole compound with high optical purity. For detailed standardized synthesis steps, please refer to the guide below.
- Complexation of divalent palladium catalyst precursor with chiral ligands such as Phox derivatives in an organic solvent.
- Reaction of alkynyl amine substrates under heating conditions with specific additives like water to facilitate cyclization.
- Post-reaction workup involving acid quenching, extraction, and purification via column chromatography to isolate high-purity products.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial benefits that translate directly into operational efficiency and cost competitiveness. The streamlined nature of the synthetic route eliminates several unit operations associated with traditional multi-step syntheses, resulting in a significantly reduced manufacturing footprint. This simplification not only lowers capital expenditure requirements for production facilities but also decreases the consumption of raw materials and utilities, contributing to a more sustainable manufacturing profile. The high selectivity of the reaction minimizes the generation of hazardous waste, simplifying compliance with environmental regulations and reducing disposal costs. Furthermore, the use of robust and commercially available catalyst components ensures a stable supply chain for critical reagents, mitigating the risk of production delays caused by material shortages. These factors collectively enhance the overall resilience of the supply chain, enabling reliable delivery of high-quality intermediates to downstream customers.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the avoidance of expensive chiral resolving agents lead to a drastic reduction in the overall cost of production. By consolidating the formation of the indole ring and the chiral center into a single catalytic event, the process maximizes resource utilization and minimizes labor costs associated with handling and processing. The high yield and selectivity of the reaction further contribute to cost savings by reducing the amount of starting material required to produce a given quantity of product. Additionally, the simplified purification process lowers the consumption of chromatography media and solvents, which are often significant cost drivers in pharmaceutical manufacturing. These cumulative efficiencies result in a more economical production model that can offer competitive pricing without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on widely available palladium catalysts and simple organic substrates ensures a secure and diversified supply base for raw materials. Unlike processes that depend on scarce natural products or proprietary reagents, this method utilizes commodity chemicals that are readily accessible from multiple vendors, reducing dependency on single sources. The robustness of the reaction conditions allows for flexible manufacturing schedules, accommodating fluctuations in demand without significant re-optimization. This stability is crucial for maintaining continuous production flows and meeting tight delivery deadlines. Moreover, the scalability of the process from gram to kilogram scales ensures that supply can be ramped up quickly to support clinical trials or commercial launch, providing partners with confidence in long-term availability.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory flasks to industrial reactors. The absence of extreme pressures or temperatures simplifies equipment requirements and enhances operational safety. From an environmental standpoint, the high atom economy and reduced waste generation align with green chemistry principles, facilitating easier regulatory approval and permitting. The minimized use of hazardous solvents and reagents lowers the environmental impact of the manufacturing process, supporting corporate sustainability goals. This alignment with environmental standards not only reduces regulatory risks but also enhances the brand reputation of the manufacturer as a responsible supplier of pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this synthesis technology. These answers are derived from the detailed experimental data and optimization studies presented in the patent documentation. They provide clarity on the scope of the method, the quality of the resulting products, and the potential for industrial application. Understanding these aspects is vital for stakeholders evaluating the integration of this technology into their existing development pipelines. The responses highlight the versatility of the catalytic system and its capacity to deliver consistent results across a range of substrate classes.
Q: What is the primary advantage of this palladium-catalyzed method over traditional indole synthesis?
A: This method efficiently constructs indole compounds containing cyano quaternary carbon chiral centers in a single catalytic step, avoiding the need for multiple protection-deprotection sequences and resolving racemic mixtures, which significantly enhances atom economy and reduces waste generation.
Q: What are the biological applications of these synthesized indole derivatives?
A: The synthesized compounds exhibit significant inhibitory activity against the Hepatitis B Virus (HBV), specifically suppressing HBV transcription, HBeAg secretion, and extracellular HBV DNA levels, making them valuable candidates for developing new antiviral therapeutics.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available palladium precursors and robust ligand systems under relatively mild conditions, allowing for scalable production with consistent enantioselectivity and simplified purification protocols suitable for industrial API intermediate supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Indole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, possessing the technical expertise to bring complex chemical innovations like this palladium-catalyzed indole synthesis to commercial reality. Our team of experienced chemists is well-versed in optimizing transition metal-catalyzed reactions for large-scale production, ensuring that the high enantioselectivity and yields observed in the laboratory are maintained at the tonnage level. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities and rigorous QC labs to guarantee stringent purity specifications for every batch. Our commitment to quality assurance means that we can deliver intermediates that meet the exacting standards required for global pharmaceutical markets, providing our partners with a reliable foundation for their drug development programs.
We invite potential collaborators to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of adopting this streamlined process for your supply chain. We encourage you to request specific COA data and route feasibility assessments to verify the suitability of these optically active indoles for your antiviral research. Together, we can accelerate the development of life-saving therapies by ensuring a steady supply of high-quality, cost-effective intermediates that drive innovation in the fight against Hepatitis B and other viral diseases.
