Revolutionizing Pharmaceutical Intermediate Synthesis: High-Efficiency Chiral Copper Catalysis for Optically Active Indole Compounds
The Chinese patent CN114160206B represents a significant advancement in asymmetric catalysis for pharmaceutical intermediate production, introducing a novel chiral copper-based catalyst system specifically designed for the high-yield synthesis of optically active indole compounds. This breakthrough technology addresses longstanding challenges in producing complex chiral molecules essential for modern drug development, with the catalyst enabling direct asymmetric alkenylation reactions that achieve remarkable enantioselectivity exceeding 91% ee and diastereoselectivity greater than 20:1. The patent demonstrates a sophisticated approach to molecular design that overcomes limitations of conventional Lewis acid catalysts, providing pharmaceutical manufacturers with a robust platform for producing high-purity indole-based intermediates critical to numerous therapeutic agents. This innovation arrives at a pivotal moment when the industry demands more efficient, sustainable methods for synthesizing complex chiral molecules while maintaining stringent quality standards required by global regulatory authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing optically active indole compounds have faced significant challenges that hinder their commercial viability for pharmaceutical manufacturing. Conventional Lewis acid catalysts typically require harsh reaction conditions, elevated temperatures, and extended reaction times to achieve modest yields and selectivity, often resulting in complex product mixtures that necessitate extensive purification processes. The poor stereoselectivity of these methods frequently leads to difficult-to-separate isomer mixtures, increasing production costs and reducing overall process efficiency. Additionally, many existing catalytic systems suffer from limited substrate scope, requiring tailored conditions for different indole derivatives, which complicates process standardization across multiple product lines. The inability to consistently achieve high enantiomeric excess (typically below 85% ee) has been a persistent limitation, forcing manufacturers to implement additional resolution steps that significantly increase production costs and reduce overall yield. Furthermore, conventional methods often require expensive transition metal catalysts that necessitate rigorous removal procedures to meet pharmaceutical purity standards, adding complexity and cost to the manufacturing process.
The Novel Approach
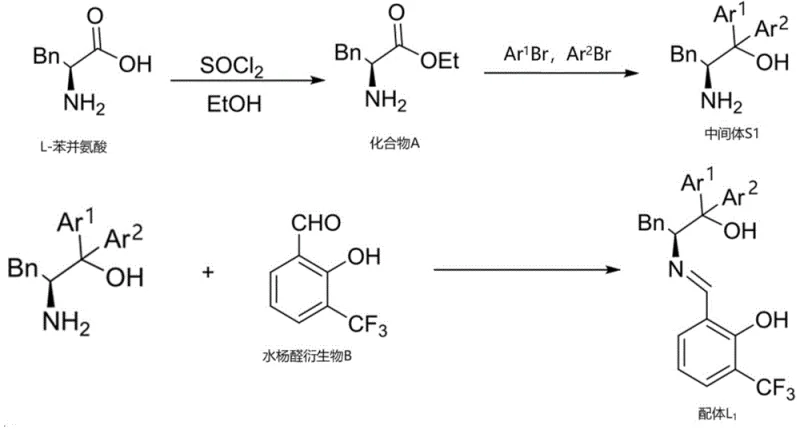
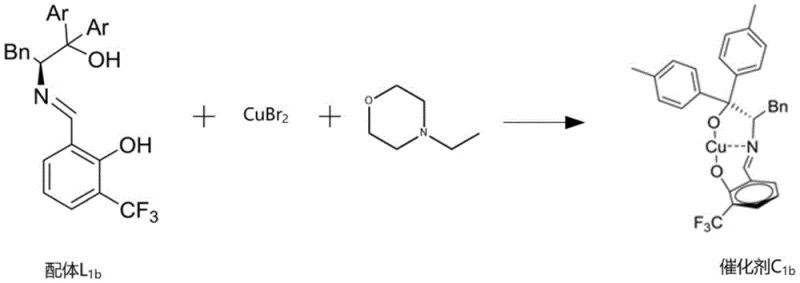
The patented chiral copper-based catalyst system represents a paradigm shift in the synthesis of optically active indole compounds by addressing these fundamental limitations through innovative molecular design. This approach utilizes specifically engineered ligands that form highly selective copper complexes capable of facilitating direct asymmetric alkenylation under mild conditions (0-25°C), achieving exceptional enantioselectivity (>91% ee) and diastereoselectivity (dr >20:1) with yields exceeding 81%. The catalyst's unique architecture creates a precisely controlled chiral environment that directs substrate approach and stabilizes key transition states, enabling high stereoselectivity without requiring extreme reaction conditions. Unlike conventional methods that often need specialized conditions for different substrates, this system demonstrates broad applicability across diverse indole derivatives while maintaining consistent performance metrics. The use of copper as the catalytic metal offers significant advantages over precious metals, including lower cost, reduced environmental impact, and simplified purification requirements that align with green chemistry principles increasingly demanded by regulatory agencies and sustainability-focused organizations.
Mechanistic Insights into Chiral Copper-Catalyzed Asymmetric Alkenylation
The exceptional performance of this catalytic system stems from its sophisticated molecular architecture, where the chiral copper complex creates a highly defined three-dimensional environment that precisely controls substrate orientation during the asymmetric alkenylation reaction. The catalyst features a carefully designed ligand framework with strategically positioned aromatic groups and functional moieties that work in concert to create optimal steric and electronic effects around the copper center. This arrangement facilitates the formation of a rigid transition state complex where the isatin-like substrate is held in a specific orientation that favors one enantioface over the other during nucleophilic attack by the 3-alkenylindole compound. The copper center acts as a Lewis acid to activate the carbonyl group of the isatin derivative while simultaneously coordinating with the ligand's nitrogen and oxygen atoms to maintain structural integrity throughout the catalytic cycle. This dual activation mechanism enables the reaction to proceed under mild conditions while achieving remarkable stereoselectivity that surpasses conventional catalytic systems by a significant margin.
The catalyst's design also incorporates critical features that minimize unwanted side reactions and impurity formation during the synthesis process. The precise spatial arrangement of substituents on the ligand framework creates a molecular pocket that excludes potential side reaction pathways while promoting the desired asymmetric transformation. This selective environment significantly reduces the formation of diastereomeric byproducts and racemic impurities that commonly plague traditional synthetic approaches. The catalyst's stability under reaction conditions prevents decomposition pathways that could lead to metal contamination or unwanted catalytic species that might compromise product purity. Furthermore, the system's ability to maintain high stereoselectivity even at elevated scales demonstrates its robustness against minor process variations, ensuring consistent product quality across different production batches. This level of impurity control is particularly valuable for pharmaceutical applications where strict regulatory requirements demand thorough understanding and control of all potential impurities.
How to Synthesize Optically Active Indole Compounds Efficiently
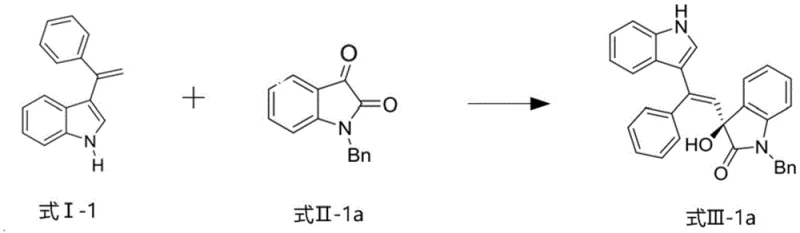
This patented methodology provides a streamlined approach to producing high-value optically active indole compounds with exceptional stereochemical control. The process begins with preparation of the chiral copper-based catalyst complex through a simple mixing procedure that combines precisely measured quantities of copper salt, organic base, and ligand under mild conditions. This catalyst system then facilitates a direct asymmetric alkenylation reaction between readily available starting materials under carefully controlled temperature conditions to produce the target compounds with remarkable selectivity. The methodology has been validated across multiple substrate combinations, demonstrating consistent performance metrics regardless of specific substituent patterns on either reaction partner. Detailed standardized synthesis steps for implementing this technology in manufacturing environments are provided below.
- Prepare the chiral copper-based catalyst complex by mixing divalent copper salt, nitrogen-containing organic base, and ligand in ethanol at room temperature for two hours, maintaining precise molar ratios to ensure optimal catalytic activity.
- Introduce the chiral copper catalyst complex to the reaction system containing the appropriate ratio of 3-alkenylindole compound and isatin-like compound, carefully controlling temperature between 0°C and 25°C to maintain stereoselectivity.
- After completion of the reaction, perform standard workup procedures including ethyl acetate extraction, saturated brine back-extraction, and column chromatography purification using petroleum ether/ethyl acetate solvent system to isolate the optically active indole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative catalytic technology delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate manufacturing. The simplified reaction pathway eliminates multiple processing steps required by conventional methods, reducing overall production complexity while enhancing yield consistency across different manufacturing scales. The elimination of expensive transition metals not only reduces raw material costs but also removes significant downstream processing requirements associated with metal removal and disposal. These combined factors create a more resilient supply chain with reduced vulnerability to raw material shortages or price volatility in specialty catalyst markets.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts significantly reduces raw material expenses while simultaneously decreasing purification complexity due to fewer side products and impurities. The mild reaction conditions lower energy consumption compared to traditional high-temperature processes, contributing to overall operational cost savings without requiring capital-intensive equipment modifications or specialized infrastructure investments.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and simple catalyst preparation procedures creates a more robust supply chain with reduced dependency on specialized or single-source suppliers. The consistent performance across different production scales ensures reliable output regardless of batch size requirements, providing greater flexibility to respond to changing market demands while maintaining product quality standards.
- Scalability and Environmental Compliance: The technology demonstrates excellent scalability from laboratory to commercial production without loss of stereoselectivity or yield, enabling seamless transition from development to manufacturing phases. The reduced environmental footprint from eliminating precious metals and minimizing waste streams aligns with increasingly stringent environmental regulations while supporting corporate sustainability initiatives without compromising process efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations regarding implementation of this patented technology in commercial pharmaceutical manufacturing environments. These answers are derived directly from the patent documentation and experimental data provided in the original filing, reflecting practical insights relevant to process development teams evaluating this methodology for potential adoption.
Q: How does this chiral copper catalyst system achieve such high enantioselectivity compared to conventional Lewis acid catalysts?
A: The chiral copper catalyst system features a precisely designed ligand framework that creates a highly stereoselective environment around the copper center, enabling asymmetric direct alkenylation with >91% ee. The unique spatial arrangement of the ligand's aromatic groups and functional moieties directs substrate approach and transition state stabilization, which conventional Lewis acids cannot replicate.
Q: What are the scalability advantages of this catalytic system for commercial pharmaceutical production?
A: This catalytic system demonstrates excellent scalability from laboratory to commercial production without loss of stereoselectivity, as evidenced by successful gram-scale reactions maintaining >90% ee. The catalyst's stability under mild reaction conditions (0-25°C) and compatibility with standard purification techniques enable seamless scale-up from kilogram to metric ton quantities while maintaining stringent quality specifications.
Q: How does this technology impact impurity profile and regulatory compliance for pharmaceutical intermediates?
A: The high stereoselectivity (>91% ee) and diastereoselectivity (dr >20:1) significantly reduce unwanted isomers and byproducts, resulting in cleaner reaction profiles with minimal impurities. This simplifies purification requirements and ensures compliance with ICH Q3 guidelines for chiral pharmaceutical intermediates, reducing regulatory risk and quality control complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Indole Compound Supplier
This patented technology represents just one example of NINGBO INNO PHARMCHEM's commitment to delivering innovative solutions for complex pharmaceutical intermediate synthesis. As a leading CDMO specialist with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise and manufacturing capabilities required to transform challenging synthetic routes into reliable commercial processes. Our stringent purity specifications and rigorous QC labs ensure consistent product quality that meets or exceeds global regulatory requirements while maintaining competitive pricing structures through optimized process design and scale-up expertise.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs. Our experts can provide detailed COA data and route feasibility assessments demonstrating how this technology can be implemented within your existing production framework while delivering significant operational improvements.
