Advanced Chiral Copper Catalyst for High-Purity Indole Pharmaceutical Intermediates
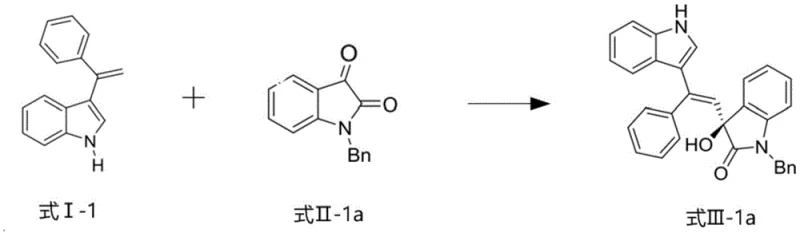
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly within the indole family, which serves as a privileged structure in numerous bioactive molecules. Patent CN114160206B introduces a groundbreaking catalytic system designed to address the longstanding challenges in synthesizing optically active 3-alkenyl-3-substituted oxindole compounds. This innovation leverages a novel chiral copper-based catalyst that facilitates direct asymmetric alkenylation of isatin-like compounds with 3-alkenylindoles. The significance of this technology lies in its ability to simultaneously achieve high enantioselectivity exceeding 91% ee and diastereoselectivity greater than 20:1 dr, while maintaining product yields above 81%. For R&D directors and process chemists, this represents a critical advancement in accessing high-purity indole derivatives essential for developing next-generation therapeutics targeting cancer, inflammation, and neurological disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quaternary carbon centers at the C3 position of oxindoles has been a formidable challenge in organic synthesis. Conventional approaches often rely on stoichiometric chiral auxiliaries or less efficient Lewis acid catalysts that struggle to differentiate between enantiotopic faces of the substrate. Previous methods, such as those utilizing cinchona alkaloids or cobalt complexes, have shown promise but frequently suffer from limited substrate scope, harsh reaction conditions, or inadequate stereocontrol when dealing with bare alkenyl groups. The poor reactivity of unsubstituted alkenyl moieties often necessitates additional protecting group manipulations or activating substituents, which adds unnecessary steps, increases waste, and complicates the purification process. These inefficiencies translate directly into higher production costs and longer lead times for high-purity indole derivatives, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
The technology disclosed in CN114160206B overcomes these barriers through a meticulously designed chiral copper catalytic system. By employing a specific Schiff base ligand derived from L-phenylalanine, the catalyst creates a highly defined chiral pocket that effectively activates the isatin substrate while controlling the approach of the 3-alkenylindole nucleophile. This direct asymmetric olefination strategy eliminates the need for pre-functionalized alkenyl reagents, streamlining the synthetic route significantly. The reaction proceeds under mild conditions, typically between 0°C and 25°C, using environmentally benign solvents like ethanol. This novel approach not only enhances the chemical efficiency with yields surpassing 81% but also ensures exceptional optical purity, making it a superior choice for cost reduction in API manufacturing where chiral integrity is paramount.
Mechanistic Insights into Chiral Copper-Catalyzed Asymmetric Alkenylation
The core of this technological breakthrough resides in the unique structure of the chiral copper catalyst, which coordinates with the substrate to enforce strict stereochemical outcomes. The catalyst is formed in situ from a divalent copper salt, a nitrogen-containing organic base, and a chiral ligand featuring a salicylaldehyde derivative moiety linked to a phenylalanine backbone. This specific arrangement allows the copper center to activate the carbonyl group of the isatin derivative through Lewis acid coordination, while the bulky aryl groups on the ligand provide steric shielding that directs the incoming nucleophile to a specific face. The resulting transition state minimizes steric clashes and maximizes electronic stabilization, leading to the observed high diastereoselectivity of dr > 20:1. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or adapt this chemistry for analogous substrates in their own pipelines.
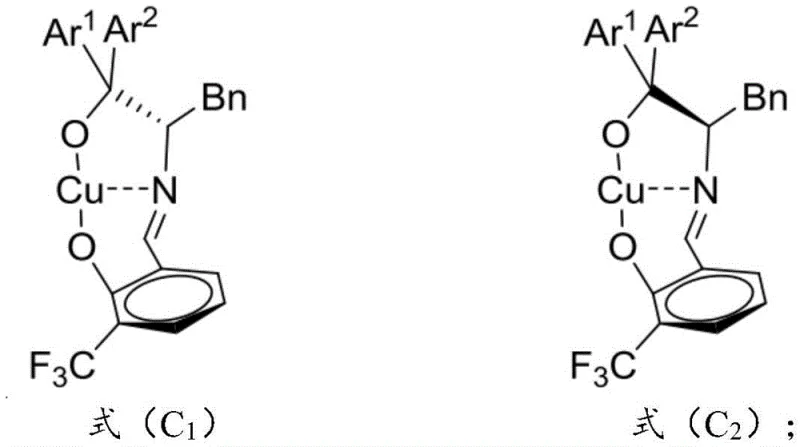
Furthermore, the impurity profile of the reaction is exceptionally clean due to the high specificity of the catalyst. The system effectively suppresses side reactions such as polymerization of the alkenyl group or non-selective background reactions that typically plague uncatalyzed or poorly catalyzed variants. The ability to maintain stereoselectivity even when scaling the reaction from milligram to gram scales indicates a robust catalytic cycle that is not easily disrupted by concentration changes or minor variations in mixing. This reliability is crucial for ensuring batch-to-batch consistency in commercial production. The catalyst's tolerance for various substituents on both the indole and isatin rings, including electron-donating and electron-withdrawing groups, further underscores its versatility. For procurement managers, this means a single catalytic platform can potentially service multiple product lines, simplifying inventory management and reducing the need for diverse catalyst sourcing.
How to Synthesize Optically Active 3-Alkenyl-3-Substituted Oxindoles Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst complex and the subsequent reaction conditions to maximize yield and purity. The process begins with the generation of the chiral ligand, followed by complexation with copper, and finally the catalytic transformation of the starting materials. The patent outlines a straightforward procedure that avoids the isolation of the catalyst intermediate, thereby reducing handling time and potential exposure to air or moisture which could degrade performance. Detailed standardized synthesis steps see the guide below, which breaks down the precise molar ratios, solvent choices, and temperature controls necessary to reproduce the high enantioselectivity reported in the patent data. Adhering to these parameters ensures the production of high-purity indole compounds that meet stringent regulatory specifications.
- Prepare the chiral copper-based catalyst complex by mixing a divalent copper salt, a specific chiral Schiff base ligand derived from phenylalanine, and a nitrogen-containing organic base in a solvent like ethanol at 0-25°C.
- Introduce the 3-alkenylindole compound and the isatin-like substrate directly into the catalyst complex mixture without isolating the catalyst, maintaining a temperature between 10-20°C for 24-48 hours.
- Perform workup by extraction with ethyl acetate, washing with saturated brine, and purifying the crude product via column chromatography to obtain the optically active oxindole with >91% ee.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that extend beyond mere chemical elegance. The elimination of expensive transition metals like palladium or rhodium in favor of copper significantly lowers the raw material costs associated with catalysis. Moreover, the high selectivity reduces the burden on downstream purification processes, as there is less need for extensive chromatography to remove diastereomeric impurities. This efficiency translates into significant cost savings in manufacturing by reducing solvent consumption, waste disposal volumes, and processing time. For supply chain heads, the use of readily available starting materials such as isatins and simple indole derivatives ensures a stable supply base, mitigating risks associated with scarce or geopolitically sensitive reagents. The robustness of the reaction conditions also implies a lower risk of batch failures, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The shift to a copper-based catalytic system inherently reduces the cost of goods sold compared to precious metal alternatives. By achieving high yields and selectivity in a single step, the process minimizes the number of unit operations required, leading to substantial cost savings. The avoidance of complex protecting group strategies further simplifies the material flow, reducing the consumption of auxiliary chemicals and lowering the overall environmental footprint of the production facility.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like ethanol and common copper salts ensures that production is not vulnerable to supply disruptions often seen with specialized reagents. The scalability of the process, demonstrated by maintained selectivity at larger scales, provides confidence in the ability to meet increasing demand without compromising quality. This stability is critical for maintaining continuous production schedules and fulfilling long-term contracts with pharmaceutical partners who require guaranteed delivery of key intermediates.
- Scalability and Environmental Compliance: The process operates under mild temperatures and uses greener solvents, aligning with modern environmental regulations and sustainability goals. The high atom economy of the direct alkenylation reaction means less waste generation per kilogram of product, simplifying waste treatment protocols. This environmental compliance not only reduces regulatory risks but also enhances the corporate social responsibility profile of the manufacturing operation, making it more attractive to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. They are derived from the specific beneficial effects and background technical problems outlined in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing frameworks. The answers reflect the proven capabilities of the system as demonstrated in the experimental examples, providing a realistic view of its performance characteristics.
Q: What are the stereochemical outcomes of this catalytic system?
A: The patented chiral copper catalyst system delivers exceptional stereocontrol, achieving enantioselectivity greater than 91% ee and diastereoselectivity exceeding a 20:1 dr ratio, ensuring high optical purity for drug candidates.
Q: How does this method improve upon conventional Lewis acid catalysis?
A: Unlike conventional Lewis acids that often struggle with reactivity and selectivity on bare alkenyl groups, this novel copper-based system utilizes a tailored chiral ligand environment to facilitate direct asymmetric alkenylation with significantly higher yields and purity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent data indicates that the stereoselectivity is maintained even when scaling up to gram-level reactions, and the use of common solvents like ethanol suggests a pathway amenable to commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Indole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral copper catalytic technology in advancing the synthesis of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of optically active indole compounds meets the highest standards required by global regulatory agencies. We are committed to leveraging such advanced patents to deliver superior value to our clients.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and secure your supply of high-value chiral intermediates.
