Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using Glucose as a Sustainable Carbon Source
Introduction to Biomass-Derived Triazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking sustainable, cost-effective pathways to access privileged heterocyclic scaffolds. Patent CN113880781A introduces a groundbreaking methodology for the preparation of 3-trifluoromethyl-substituted 1,2,4-triazole compounds, utilizing glucose as a renewable carbon source. This innovation addresses critical challenges in modern organic synthesis by replacing expensive, petrochemical-derived aldehydes with abundant biomass. The trifluoromethyl triazole motif is a cornerstone structure in medicinal chemistry, found in blockbuster drugs such as Sitagliptin and various kinase inhibitors, due to its ability to enhance metabolic stability and lipophilicity. By leveraging a trifluoromethanesulfonic acid-catalyzed cascade reaction, this process achieves high efficiency under mild thermal conditions, offering a robust alternative for the production of high-purity pharmaceutical intermediates.
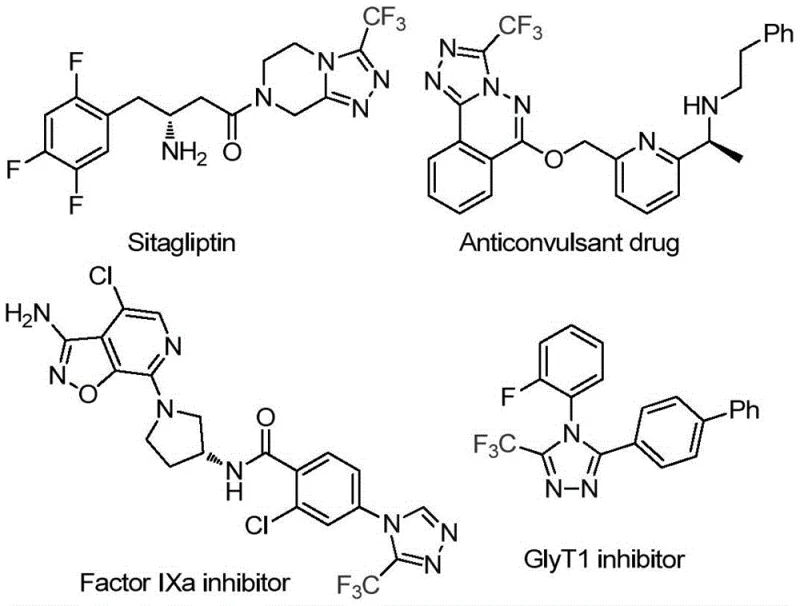
The significance of this technology extends beyond mere academic interest; it represents a tangible shift towards green chemistry in industrial manufacturing. The protocol operates effectively at temperatures between 70°C and 90°C, utilizing tert-butyl hydroperoxide (TBHP) as a terminal oxidant to drive the aromatization step. For R&D directors and process chemists, this method offers a streamlined route that minimizes waste and maximizes atom economy. The ability to generate diverse substitution patterns on the N-1 position of the triazole ring further enhances its utility for lead optimization campaigns, making it a highly attractive candidate for integration into existing synthetic pipelines for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 3-trifluoromethyl-1,2,4-triazoles often rely on the condensation of trifluoroacetamidine derivatives with specific aldehydes or carboxylic acid derivatives. These conventional pathways frequently suffer from significant drawbacks, including the requirement for harsh reaction conditions, such as high temperatures or strong bases, which can compromise functional group tolerance. Furthermore, many established methods necessitate strictly anhydrous and anaerobic environments to prevent side reactions or decomposition of sensitive intermediates, thereby increasing operational complexity and infrastructure costs. The reliance on pre-functionalized aldehyde starting materials also introduces supply chain vulnerabilities, as these reagents can be costly and subject to market volatility. Additionally, the use of stoichiometric amounts of heavy metal oxidants or hazardous dehydrating agents in older protocols generates substantial toxic waste, posing environmental compliance challenges for large-scale production facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes D-glucose, a ubiquitous and inexpensive carbohydrate, as the primary carbon synthon. This biomass-derived strategy fundamentally alters the economic and environmental profile of the synthesis. The reaction proceeds via an acid-catalyzed cleavage of glucose to generate reactive aldehyde species in situ, which immediately undergo condensation with trifluoroethylimide hydrazide. This tandem process eliminates the need for isolating unstable aldehyde intermediates and removes the dependency on external aldehyde sourcing. The use of trifluoromethanesulfonic acid (TfOH) as a catalyst allows the reaction to proceed under relatively mild conditions without the need for inert atmosphere protection. This operational simplicity translates directly to reduced capital expenditure on specialized reactor equipment and lower energy consumption, positioning this method as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Cascade Cyclization
The core of this transformation lies in a sophisticated cascade mechanism initiated by the strong Brønsted acid catalyst, trifluoromethanesulfonic acid. Initially, the acid promotes the cleavage of the glycosidic bonds in glucose, effectively cracking the sugar molecule to release reactive aldehyde fragments. These in situ generated aldehydes then engage in a condensation reaction with the hydrazine moiety of the trifluoroethylimide hydrazide substrate, forming a hydrazone intermediate. This step is crucial as it sets the stage for the subsequent ring closure. Following hydrazone formation, an intramolecular nucleophilic addition occurs, where the nitrogen atom attacks the electrophilic center, leading to the formation of the five-membered triazoline ring system. This cyclization is driven by the electronic activation provided by the trifluoromethyl group and the acidic medium.
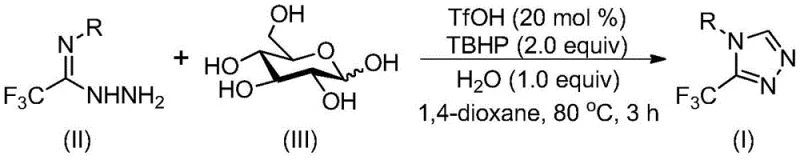
The final and critical step involves the aromatization of the triazoline intermediate to yield the stable 1,2,4-triazole product. This oxidative aromatization is facilitated by tert-butyl hydroperoxide (TBHP), which acts as a green oxidant, accepting electrons to restore aromaticity to the heterocyclic ring. The presence of water in the reaction mixture, rather than being detrimental, actually aids in the hydrolysis steps required for glucose degradation, demonstrating the robustness of the system. From an impurity control perspective, this mechanism is highly favorable because the in situ generation of the aldehyde prevents the accumulation of free aldehyde impurities that could otherwise lead to polymerization or side reactions. The specificity of the acid catalyst ensures that the cyclization occurs regioselectively, minimizing the formation of isomeric byproducts and simplifying downstream purification processes.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazoles Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-scale production. The protocol involves a straightforward one-pot procedure where all reagents are combined in a suitable organic solvent, such as 1,4-dioxane, which has been identified as optimal for solubility and reaction rate. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and safety.
- Combine trifluoroethylimide hydrazide, glucose, trifluoromethanesulfonic acid (catalyst), TBHP (oxidant), and water in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 70-90°C and stir for 2-4 hours to allow acid-catalyzed cleavage of glucose and subsequent cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this glucose-based synthesis offers compelling economic and logistical benefits. The primary driver for cost optimization is the replacement of expensive, specialty aldehyde reagents with glucose, a commodity chemical available in massive quantities globally at a fraction of the cost. This shift significantly lowers the raw material bill of materials (BOM), directly impacting the gross margin of the final API. Furthermore, the elimination of stringent anhydrous and anaerobic requirements reduces the need for specialized drying agents, inert gases, and complex reactor setups, leading to substantial operational expenditure (OpEx) savings. The simplified workup procedure, which typically involves filtration and standard chromatography, minimizes solvent usage and processing time, enhancing overall throughput.
- Cost Reduction in Manufacturing: The utilization of glucose as a carbon source drastically reduces raw material costs compared to traditional petrochemical aldehydes. By avoiding the use of precious metal catalysts and expensive dehydrating agents, the process achieves significant cost efficiency. The mild reaction conditions also translate to lower energy consumption for heating and cooling, further driving down the manufacturing cost per kilogram. This economic advantage allows for more competitive pricing strategies in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Glucose is a biomass feedstock with a highly stable and diversified global supply chain, immune to the geopolitical fluctuations often associated with petrochemical derivatives. This ensures a consistent and reliable supply of starting materials, mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without requiring extensive re-validation of sensitive parameters, ensuring business continuity.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable from gram to multi-gram levels with high efficiency, indicating strong potential for ton-scale commercial production. The use of water as an additive and TBHP as an oxidant aligns with green chemistry principles, reducing the generation of hazardous waste streams. This environmental compatibility simplifies regulatory compliance and waste disposal logistics, making it an ideal candidate for sustainable manufacturing initiatives demanded by modern ESG (Environmental, Social, and Governance) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucose-mediated triazole synthesis. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of using glucose in this synthesis?
A: Glucose acts as a renewable, low-cost biomass carbon source that eliminates the need for expensive aldehyde precursors. It simplifies the supply chain and reduces raw material costs significantly compared to traditional petrochemical routes.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the major operational benefits of this patented method is that it proceeds efficiently without the need for rigorous anhydrous or oxygen-free environments, greatly simplifying reactor requirements and operational complexity.
Q: What is the substrate scope for the R group in the triazole product?
A: The method demonstrates broad compatibility, successfully synthesizing derivatives where R is phenethyl or various substituted aryl groups (including methyl, methoxy, halogen, and methylthio substituents), allowing for diverse library generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biomass-derived synthetic routes in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this glucose-based triazole synthesis can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of acid-catalyzed cascade reactions, maintaining stringent purity specifications through our rigorous QC labs to guarantee the quality of every batch delivered.
We invite you to collaborate with us to leverage this cost-effective and sustainable technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with high-purity 3-trifluoromethyl-1,2,4-triazoles.
