Advanced Synthesis of 3-Trifluoromethyl Triazoles: Enabling Cost-Efficient Commercial Scale-Up for High-Purity Pharmaceutical Intermediates
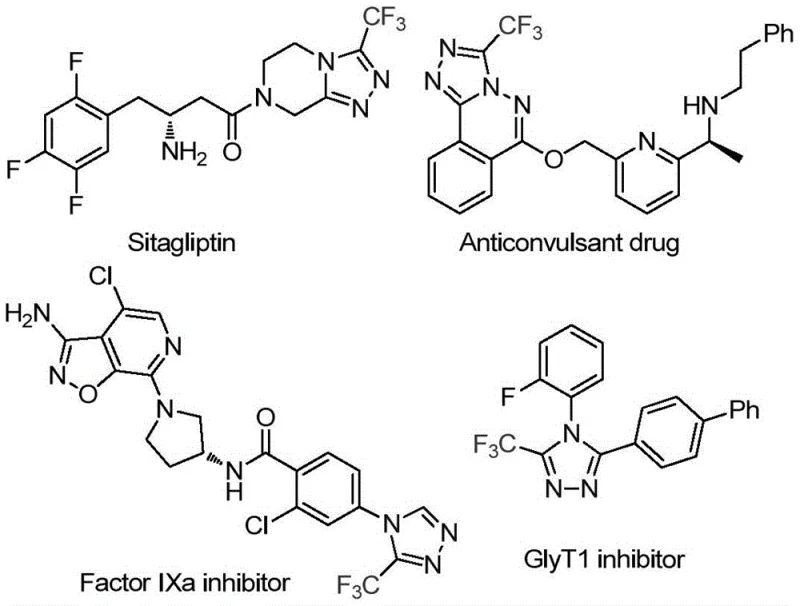
Patent CN114920707B introduces a transformative methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds—a critical class of heterocyclic molecules with extensive applications in pharmaceutical development due to their prevalence in bioactive scaffolds such as Sitagliptin and GlyT1 inhibitors. This innovative process leverages dimethylformamide (DMF) as both solvent and carbon source under ambient atmospheric conditions, eliminating the need for stringent anhydrous or anaerobic environments that complicate conventional synthetic routes while maintaining high reaction efficiency across diverse substrates. The methodology demonstrates remarkable operational simplicity with reaction parameters optimized for industrial implementation at temperatures between 110°C and 130°C over a duration of 10 to 15 hours using molecular iodine as a promoter. By utilizing commercially available starting materials and avoiding transition metal catalysts, this approach achieves significant process intensification while accommodating strategic molecular design to produce triazole derivatives with tailored functional groups. The resulting compounds exhibit exceptional potential as core building blocks in next-generation therapeutics targeting metabolic disorders and neurological conditions, representing a substantial advancement in heterocyclic chemistry that directly addresses pharmaceutical manufacturers' demands for robust intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of trifluoromethyl-substituted triazoles often requires transition metal catalysts under strictly anhydrous and anaerobic conditions that necessitate specialized equipment and rigorous environmental controls, significantly increasing operational complexity and capital expenditure while introducing potential metal contamination risks in final products. These methods typically employ multi-step sequences with low functional group tolerance that limit substrate diversity and necessitate extensive purification procedures to remove residual metals or byproducts that compromise purity specifications required for pharmaceutical intermediates. The reliance on expensive catalysts such as palladium or copper creates substantial cost barriers at commercial scale while generating hazardous waste streams that complicate environmental compliance and increase disposal costs. Furthermore, conventional approaches frequently suffer from narrow substrate scope where sensitive functional groups require protection/deprotection steps that reduce overall yield and extend production timelines. The stringent reaction conditions also impose significant scalability challenges when transitioning from laboratory to manufacturing environments due to heat transfer limitations and safety concerns associated with oxygen-sensitive reagents.
The Novel Approach
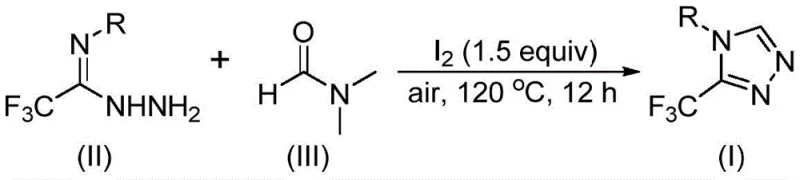
The patented methodology overcomes these limitations through an iodine-promoted tandem cyclization process that operates efficiently under ambient air atmosphere without requiring moisture-sensitive reagents or specialized containment systems. By utilizing DMF as both solvent and carbon source—where its methyl and formyl groups participate directly in the reaction mechanism—the process eliminates the need for external carbon donors while enabling simplified workup procedures that avoid complex metal removal steps. This approach demonstrates exceptional substrate flexibility accommodating aryl groups with diverse substituents including halogens, alkyl groups, alkoxy moieties, and trifluoromethyl functionalities across various positional isomers without requiring protective group strategies. The reaction achieves high conversion rates at moderate temperatures (120°C) within practical timeframes (12 hours), making it readily adaptable to standard manufacturing equipment without significant capital investment. Crucially, the elimination of transition metals not only reduces raw material costs but also streamlines quality control by removing stringent metal residue testing requirements that typically delay product release in pharmaceutical manufacturing.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The reaction proceeds through two distinct mechanistic pathways depending on which component of DMF serves as the carbon source, both initiated by molecular iodine activation that facilitates the critical C-N bond formation required for triazole ring construction. When DMF's formyl group acts as carbon donor, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate that subsequently undergoes intramolecular cyclization followed by dimethylamine elimination to yield the triazole product—this pathway benefits from DMF's dual role as solvent that maintains optimal concentration for cyclization kinetics. Alternatively, when the N-methyl group participates as carbon source, iodine first generates an amine salt from DMF that eliminates hydrogen iodide before nucleophilic addition to the hydrazide; this forms an azadiene intermediate that undergoes intramolecular addition followed by oxidative aromatization to produce the final triazole structure. Both pathways operate efficiently under identical reaction conditions due to DMF's unique molecular architecture that provides multiple reactive sites while maintaining solubility throughout the transformation sequence.
Impurity control is achieved through the inherent selectivity of the iodine-promoted cyclization where the reaction conditions favor exclusive formation of the desired triazole regioisomer without significant byproduct generation; this selectivity stems from the precise steric and electronic requirements of the cyclization transition state that disfavors alternative ring-closure pathways. The absence of transition metals eliminates common impurities such as metal complexes or ligand-derived contaminants that typically require additional purification steps in conventional syntheses. Furthermore, the moderate reaction temperature (120°C) prevents thermal decomposition pathways that could generate degradants while the air atmosphere avoids over-reduction side reactions observed in anaerobic systems. Post-reaction workup through standard column chromatography effectively removes minor impurities such as unreacted starting materials or dimethylamine byproducts without requiring specialized techniques—this straightforward purification protocol ensures consistent production of high-purity intermediates meeting stringent pharmaceutical quality standards.
How to Synthesize 3-Trifluoromethyl Triazoles Efficiently
This patented methodology provides a streamlined pathway for producing high-purity trifluoromethyl-substituted triazoles through a single-step iodine-promoted cyclization that eliminates multiple intermediate isolation steps required in traditional approaches while maintaining exceptional functional group tolerance across diverse substrates. The process leverages commercially available starting materials—molecular iodine as promoter and DMF as dual-function solvent/reactant—that simplify supply chain logistics compared to methods requiring specialized reagents or catalysts. Reaction optimization studies confirm robust performance across temperature ranges from 110°C to 130°C with consistent yields achieved within standard manufacturing timeframes of 10–15 hours under ambient air conditions without requiring inert gas handling systems. Detailed standardized synthesis procedures are provided below to enable seamless technology transfer from laboratory development to commercial manufacturing environments while ensuring reproducible quality outcomes.
- Combine molecular iodine (1.5 equivalents), trifluoroethyliminohydrazide substrate (R = aryl), and excess DMF solvent in a reaction vessel under air atmosphere without requiring anhydrous or anaerobic environments.
- Heat the mixture to 120°C and maintain for 12 hours to ensure complete conversion through tandem cyclization where DMF serves as both solvent and carbon source.
- Perform standard workup including filtration, washing, silica gel mixing, and column chromatography purification to obtain high-purity triazole products with diverse functional group tolerance.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by transforming complex multi-step processes into a single-operation transformation that significantly reduces operational complexity while enhancing supply chain resilience through simplified material requirements and reduced environmental dependencies. The elimination of transition metal catalysts not only removes costly reagent expenses but also eliminates associated waste treatment requirements that typically create bottlenecks in production scheduling and increase regulatory compliance burdens across global manufacturing sites. By operating effectively under standard atmospheric conditions without specialized containment systems, this approach substantially lowers capital expenditure barriers for new production facilities while enabling faster technology transfer between existing manufacturing locations through standardized equipment requirements.
- Cost Reduction in Manufacturing: The dual functionality of DMF as both solvent and carbon source eliminates separate reagent procurement while avoiding expensive metal catalysts and their associated removal processes; this integrated approach significantly reduces raw material costs and simplifies waste stream management by generating fewer byproducts that require specialized disposal protocols.
- Enhanced Supply Chain Reliability: Utilizing commercially ubiquitous materials like molecular iodine and DMF ensures consistent availability from multiple global suppliers while eliminating dependencies on scarce or geopolitically sensitive catalysts; the atmospheric reaction conditions further enhance reliability by removing vulnerability to inert gas supply chain disruptions.
- Scalability and Environmental Compliance: The straightforward process design enables seamless scale-up from laboratory to commercial production without reoptimization due to consistent performance across temperature ranges; simplified workup procedures generate less hazardous waste compared to metal-catalyzed alternatives while maintaining compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this patented technology for integration into their supply chains; responses are derived directly from experimental data presented in patent CN114920707B regarding process robustness and commercial applicability across diverse manufacturing environments.
Q: How does this method eliminate the need for anhydrous/anaerobic conditions compared to conventional triazole synthesis?
A: The process utilizes DMF as both solvent and carbon source under ambient air atmosphere, avoiding moisture-sensitive catalysts or oxygen-sensitive intermediates that typically require specialized equipment in traditional methods.
Q: What substrate diversity can be achieved with this iodine-promoted cyclization approach?
A: The methodology accommodates broad substrate scope including aryl groups with electron-donating (methyl, methoxy), electron-withdrawing (halogen, trifluoromethyl), and sterically hindered substituents across ortho/meta/para positions.
Q: How does the dual role of DMF enhance commercial viability for pharmaceutical manufacturers?
A: By serving as both reaction medium and carbon source, DMF eliminates additional reagents while enabling simplified workup procedures that reduce operational complexity and associated costs in large-scale production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Triazole Supplier
Our company leverages this patented technology to deliver high-purity triazole intermediates through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through advanced analytical capabilities in our ISO-certified QC labs. As a leading CDMO partner specializing in complex heterocyclic synthesis, we combine deep expertise in iodine-promoted cyclization chemistry with robust manufacturing infrastructure to ensure consistent supply chain performance regardless of volume requirements or regulatory jurisdiction.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your production needs—contact us today to discuss how our patented methodology can enhance your pharmaceutical intermediate supply strategy.
