Scalable Photochemical Synthesis of High-Purity Phenanthrene Derivatives for Advanced Materials
Scalable Photochemical Synthesis of High-Purity Phenanthrene Derivatives for Advanced Materials
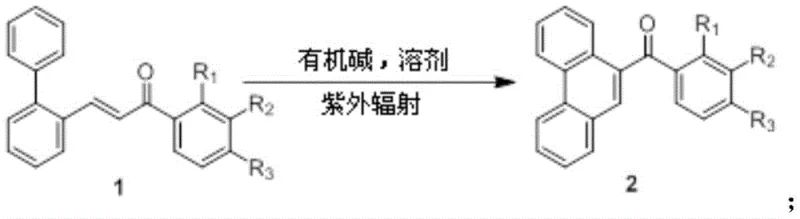
The rapid evolution of the organic electronics and pharmaceutical sectors demands increasingly sophisticated synthetic methodologies that balance high purity with environmental sustainability. Patent CN113443974A introduces a groundbreaking approach for the preparation of phenanthrene derivatives, a class of fused-ring aromatic hydrocarbons critical for applications ranging from OLED emissive layers to specialized pharmaceutical intermediates. This technology leverages a metal-free, photo-induced intramolecular cyclization strategy that fundamentally shifts the paradigm from traditional transition-metal catalysis to a greener, more efficient photocatalytic process. By utilizing biphenyl ketene as a key starting material and employing ultraviolet radiation in the presence of an organic base, this method achieves high conversion rates while eliminating the heavy metal contamination risks associated with conventional palladium-catalyzed routes. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline supply chains for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the phenanthrene nucleus has relied heavily on transition-metal catalyzed cycloisomerization or oxidative cyclization processes involving aromatic hydrocarbons and ortho-alkynyl-biaryls. While effective, these traditional pathways often necessitate the use of toxic and volatile halogenated reagents for substrate preparation, such as the bromination of chalcone substrates, which introduces significant operational complexity and safety hazards. Furthermore, the reliance on heavy metal catalysts like palladium creates a substantial downstream burden; removing trace metal residues to meet the stringent purity specifications required for electronic materials or APIs involves costly purification steps, including scavenger resins and repeated chromatography. The environmental footprint of these methods is considerable, generating heavy metal waste that complicates disposal and increases the overall cost of goods sold, making them less attractive for large-scale commercial manufacturing in a regulated environment.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct intramolecular cyclization of biphenyl ketene driven by ultraviolet light, completely bypassing the need for transition metal catalysts in the ring-closing step. This photochemical activation allows hydrogen electrons on the double bond of the biphenyl ketene to transition to an active state, facilitating a cyclization reaction with the adjacent benzene ring to form the aliphatic intermediate, which is subsequently aromatized by the organic base. This approach not only simplifies the reaction setup by operating under mild conditions (room temperature, atmospheric pressure) but also drastically reduces the impurity profile by avoiding metal leaching. The use of common organic bases like DBU or triethylamine further enhances the economic viability, as these reagents are inexpensive and easy to handle compared to sensitive organometallic complexes, offering a robust alternative for the cost reduction in electronic chemical manufacturing.
Mechanistic Insights into UV-Induced Intramolecular Cyclization
The core of this technological breakthrough lies in the precise manipulation of electronic states through photon absorption. Under the excitation of ultraviolet light, typically provided by a 20-40W lamp, the biphenyl ketene molecule absorbs energy, promoting electrons from the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO). This excitation renders the olefinic double bond highly reactive, enabling a 6-pi electrocyclization or a radical-mediated cyclization pathway that fuses the biaryl system into the rigid phenanthrene skeleton. Simultaneously, the presence of oxygen from the air acts as a mild oxidant, assisting in the re-aromatization process, while the organic base serves a dual role: it neutralizes acidic byproducts and actively participates in the deprotonation of the dihydro-intermediate to restore aromaticity. This synergistic effect between photo-excitation and base catalysis ensures high selectivity and minimizes side reactions such as polymerization or over-oxidation, which are common pitfalls in thermal cyclization methods.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity phenanthrene derivatives. Since the reaction does not involve metal-ligand coordination spheres, there is no risk of forming metal-complexed impurities that are notoriously difficult to separate. The primary byproducts are typically limited to unreacted starting materials or minor isomeric forms, which can be easily removed via standard crystallization or flash chromatography. The patent data indicates that substituents such as chlorine, bromine, iodine, or nitro groups on the aromatic rings are well-tolerated, suggesting a broad substrate scope that maintains high regioselectivity. This robustness is critical for R&D teams aiming to synthesize diverse libraries of functionalized phenanthrenes for structure-activity relationship (SAR) studies or material property tuning without compromising on batch-to-batch consistency.
How to Synthesize Phenanthrene Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the preparation of the biphenyl ketene precursor, which itself is synthesized through a two-step sequence involving a Suzuki coupling followed by an aldol condensation, ensuring high quality of the starting material before the critical cyclization step. Once the precursor is obtained, it is dissolved in a suitable solvent such as chlorobenzene or toluene at a concentration of 0.1-0.25 mmol/mL, and the organic base is added in a molar excess to drive the equilibrium forward. The detailed standardized synthesis steps, including specific workup procedures and purification techniques to achieve pharmaceutical grade purity, are provided in the guide below.
- Prepare the biphenyl ketene precursor via Suzuki coupling and aldol condensation.
- Dissolve the biphenyl ketene in chlorobenzene or toluene with an organic base like DBU.
- Irradiate the mixture with a 20-40W UV lamp under air for 4-8 hours to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical route translates into tangible strategic benefits beyond mere technical feasibility. The elimination of transition metal catalysts in the final cyclization step removes a major bottleneck in the supply chain, as it negates the need for sourcing expensive palladium salts and the associated logistics of handling hazardous heavy metal waste. This simplification leads to substantial cost savings in raw material procurement and waste disposal, directly improving the margin profile of the final product. Moreover, the use of air as a co-oxidant and common solvents like ethanol or chlorobenzene ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents, enhancing overall supply security for long-term contracts.
- Cost Reduction in Manufacturing: The absence of noble metal catalysts significantly lowers the direct material costs, while the simplified post-treatment process reduces the consumption of silica gel and solvents required for metal scavenging. By avoiding complex purification protocols needed to meet low residual metal limits, manufacturers can achieve faster batch turnover times and lower utility consumption, resulting in a more competitive pricing structure for high-purity phenanthrene derivatives without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, commodity-grade chemicals such as organic bases and standard solvents mitigates the risk of supply disruptions often associated with specialized catalytic systems. The robustness of the photochemical reaction conditions allows for flexible manufacturing scheduling, as the reaction can be easily monitored and controlled using standard UV equipment, ensuring consistent delivery timelines and reducing lead time for high-purity phenanthrene derivatives even during periods of high market demand.
- Scalability and Environmental Compliance: The process is inherently scalable, as UV reactors can be readily adapted for continuous flow chemistry or larger batch vessels, facilitating the commercial scale-up of complex electronic chemicals. Furthermore, the green chemistry attributes of this method, including reduced toxicity and lower E-factor due to the absence of heavy metals, align perfectly with increasingly stringent global environmental regulations, minimizing the regulatory burden and potential liability for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocyclization technology, based on the specific data and embodiments provided in the patent documentation. These insights are designed to clarify the operational parameters and potential advantages for stakeholders evaluating this synthetic route for their specific applications.
Q: Does this photocyclization process require expensive transition metal catalysts?
A: No, the core cyclization step described in patent CN113443974A utilizes ultraviolet radiation and an organic base (such as DBU) instead of palladium or other heavy metal catalysts, significantly simplifying purification and reducing environmental impact.
Q: What is the typical yield for the phenanthrene derivative synthesis?
A: According to the experimental examples in the patent, the process achieves high conversion rates with isolated yields ranging typically from 60% to 88%, depending on the specific substituents on the aromatic rings.
Q: Can this method be scaled for industrial production of OLED materials?
A: Yes, the method uses standard solvents like chlorobenzene and common UV lamps (20-40W), making it highly suitable for commercial scale-up of complex electronic chemicals without the need for specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenanthrene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free photochemical synthesis in advancing the production of next-generation organic materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phenanthrene derivative delivered meets the exacting standards required for OLED displays and pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical high-value intermediates.
