Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Manufacturing
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN112480015B, which discloses a robust multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This class of compounds is of paramount importance in medicinal chemistry due to the widespread presence of the quinazolinone core in numerous bioactive molecules, ranging from antifungal and antibacterial agents to anticancer drugs. The introduction of a trifluoromethyl group further enhances the metabolic stability and lipophilicity of these drug candidates, making them highly desirable targets for R&D teams globally. The patented methodology offers a streamlined approach that bypasses many of the logistical and safety hurdles associated with traditional carbonylation reactions.
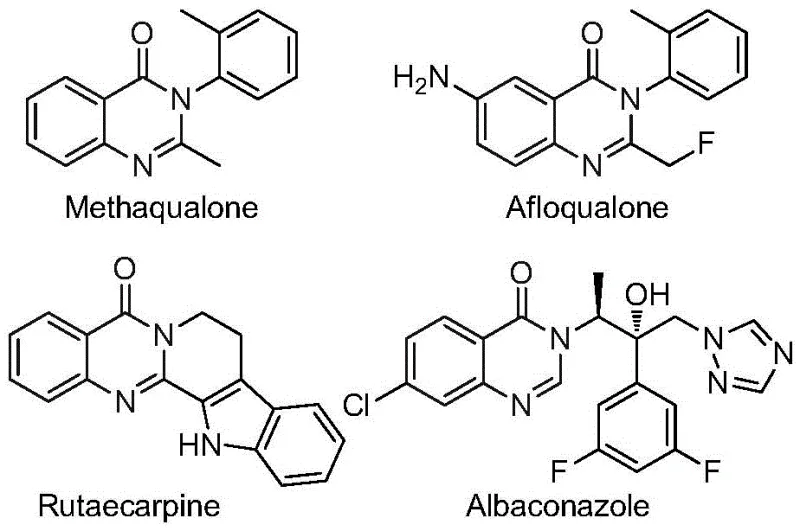
Quinazolinone compounds represent a critical structural motif found in a vast array of functional molecules and approved pharmaceuticals. As illustrated in the structural diversity of known drugs, the ability to efficiently construct this fused nitrogen-containing six-membered heterocycle is a valuable capability for any fine chemical manufacturer. The specific incorporation of the trifluoromethyl moiety is not merely a structural variation but a strategic modification that significantly improves the physicochemical properties of the parent molecule, such as electronegativity and bioavailability. Consequently, developing a synthesis that can reliably install this group while maintaining the integrity of the quinazolinone core is a high priority for reliable pharmaceutical intermediate suppliers aiming to support drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazolinone rings, particularly those bearing trifluoromethyl substituents, has been plagued by significant synthetic challenges that hinder scalability and cost-efficiency. Conventional strategies often rely on the use of nitro compounds, which are indeed cheap and accessible, but the methods to convert them into the desired heterocycles are frequently problematic. For instance, traditional ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides typically requires high-pressure carbon monoxide conditions, posing severe safety risks and requiring specialized high-pressure reactor infrastructure that is not universally available. Other methods involve iron-catalyzed condensations or palladium-catalyzed cyclizations that necessitate expensive, pre-activated substrates like 2-bromoformylaniline or acid anhydrides. These precursors not only drive up the raw material costs but also generate substantial amounts of waste during their own preparation, negatively impacting the overall atom economy and environmental footprint of the process.
The Novel Approach
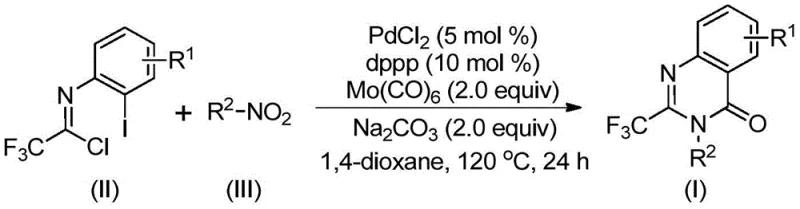
In stark contrast to these cumbersome legacy techniques, the method described in patent CN112480015B introduces a transformative one-pot strategy that utilizes transition metal palladium catalysis to couple trifluoroethylimidoyl chloride directly with nitro compounds. This novel approach elegantly circumvents the need for hazardous high-pressure CO gas by employing molybdenum hexacarbonyl (Mo(CO)6) as a solid, easy-to-handle carbon monoxide surrogate. The reaction proceeds under relatively mild thermal conditions in a standard organic solvent, eliminating the requirement for complex pre-activation steps. By merging the reduction of the nitro group, the formation of the carbon-nitrogen bond, and the carbonylation cascade into a single operational sequence, this method drastically simplifies the workflow. This simplification is a key driver for cost reduction in pharmaceutical intermediate manufacturing, as it reduces labor hours, minimizes solvent usage, and lowers the barrier to entry for producing these high-value scaffolds.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors focused on process optimization and impurity control. The reaction is believed to initiate with the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which serves a dual role as both the CO source and the reducing agent. Following this in situ generation of the amine, a base-promoted intermolecular coupling occurs between the amine and the trifluoroethylimidoyl chloride, yielding a trifluoroacetamidine derivative. The palladium catalyst then inserts into the carbon-iodine bond of the aromatic ring to form a divalent palladium intermediate. Simultaneously, the thermal decomposition of Mo(CO)6 releases carbon monoxide, which inserts into the carbon-palladium bond to generate an acyl-palladium species. This intricate dance of organometallic steps culminates in the formation of a seven-membered cyclic palladium intermediate, which subsequently undergoes reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product.
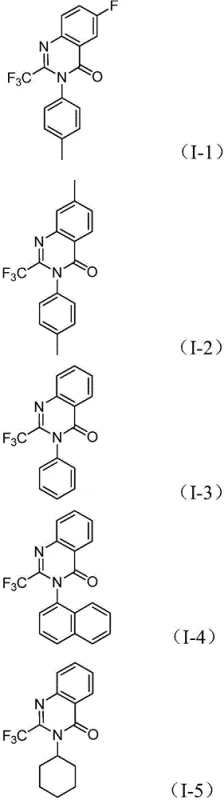
From a quality control perspective, the elegance of this mechanism lies in its convergence. By generating the reactive amine species in situ, the process avoids the isolation of potentially unstable intermediates that could degrade or form side products. Furthermore, the use of a specific ligand system, typically 1,3-bis(diphenylphosphino)propane (dppp), ensures high regioselectivity and suppresses the formation of homocoupling byproducts often seen in palladium chemistry. The compatibility of this catalytic system with a wide range of functional groups—such as halogens, alkyls, and alkoxy groups on the aromatic rings—means that the impurity profile remains manageable even when synthesizing diverse analogs. This robustness is essential for ensuring high-purity pharmaceutical intermediates that meet the stringent specifications required for downstream drug development.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The execution of this synthesis is designed to be straightforward, leveraging commercially available reagents to minimize supply chain friction. The protocol involves charging a reaction vessel with the palladium catalyst, the dppp ligand, sodium carbonate as the base, and the solid CO source Mo(CO)6. To this mixture, the key building blocks—trifluoroethylimidoyl chloride and the chosen nitro compound—are added in an aprotic organic solvent such as 1,4-dioxane. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometric ratios and thermal profiles necessary to achieve optimal yields. This operational simplicity makes the method highly attractive for both laboratory-scale discovery and pilot-plant operations.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards using nitro compounds as primary feedstocks represents a significant opportunity for cost optimization, as these materials are commodity chemicals with stable global supply chains, unlike specialized brominated or iodinated precursors which can be subject to price volatility and long lead times. Furthermore, the elimination of high-pressure gas cylinders removes a major safety liability and logistical burden from the manufacturing site, simplifying regulatory compliance and insurance overheads. The ability to run this reaction in a one-pot fashion also translates to reduced solvent consumption and waste generation, aligning with modern green chemistry initiatives and lowering disposal costs.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the substitution of expensive, pre-functionalized starting materials with cheap nitroarenes and the use of a solid CO source. By avoiding the multi-step synthesis of activated benzamides or anhydrides, the overall step count is reduced, which inherently lowers the cost of goods sold (COGS). Additionally, the high reaction efficiency and yield reported across various substrates mean that less raw material is wasted, further enhancing the economic viability of the process for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on widely available nitro compounds and standard palladium catalysts mitigates the risk of supply disruptions that often plague specialty chemical markets. Since the reagents are not bespoke or difficult to synthesize, sourcing can be diversified across multiple vendors, ensuring continuity of supply. This reliability is critical for maintaining production schedules and meeting the demanding delivery timelines of international pharmaceutical clients who require consistent quality and volume.
- Scalability and Environmental Compliance: The use of Mo(CO)6 as a CO surrogate significantly improves the safety profile of the reaction, making it easier to scale from gram to kilogram quantities without the need for specialized high-pressure infrastructure. This facilitates faster technology transfer from R&D to production. Moreover, the cleaner reaction profile and reduced waste generation simplify the effluent treatment process, helping manufacturers meet increasingly strict environmental regulations regarding volatile organic compounds and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific advantages and operational details outlined in the patent documentation. These insights are intended to clarify the feasibility and benefits of implementing this route in a commercial setting.
Q: What are the key advantages of using nitro compounds in this synthesis?
A: Nitro compounds serve as inexpensive and readily available starting materials that replace costly pre-activated substrates, significantly reducing raw material costs while maintaining high reaction efficiency.
Q: How does this method improve safety compared to traditional carbonylation?
A: By utilizing solid Mo(CO)6 as an in situ carbon monoxide source, the process eliminates the need for handling high-pressure toxic CO gas, thereby enhancing operational safety and simplifying equipment requirements.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the method demonstrates excellent substrate compatibility and has been validated for gram-scale expansion, indicating strong potential for commercial scale-up with robust impurity control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable industrial supply. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed transformations allows us to optimize this specific quinazolinone synthesis for maximum yield and minimal impurity levels, providing our partners with a competitive edge in the market.
We invite potential collaborators to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data for related compounds and to schedule a consultation for comprehensive route feasibility assessments, ensuring your supply chain is built on the most efficient and robust chemistry available.
