Revolutionizing Pharmaceutical Intermediate Production with Efficient Trifluoromethyl Quinazolinone Synthesis Technology
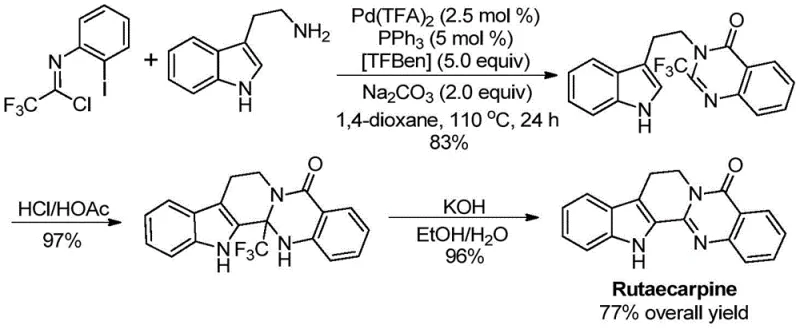
The patent CN113045503A introduces a groundbreaking methodology for synthesizing structurally diverse trifluoromethyl-substituted quinazolinone compounds through a palladium-catalyzed carbonylation process that addresses critical limitations in current pharmaceutical intermediate manufacturing techniques This innovative approach leverages readily accessible starting materials including trifluoroethylimidoyl chloride and various amines to construct complex heterocyclic frameworks essential for drug development The process operates under precisely controlled conditions at 110°C for durations between 16 to 30 hours achieving remarkable efficiency without requiring specialized equipment or hazardous reagents This breakthrough represents a significant advancement over conventional synthetic routes by eliminating pre-activation steps and enabling direct access to high-value intermediates used in multiple therapeutic agents The methodology's robustness has been validated through successful application in synthesizing Rutaecarpine a bioactive natural product demonstrating its practical utility in real-world pharmaceutical production scenarios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for trifluoromethyl quinazolinones typically involve cyclization reactions using anthranilamide derivatives with ethyl trifluoroacetate or trifluoroacetic anhydride under harsh conditions that often require elevated temperatures exceeding safe operational limits or prolonged reaction times leading to significant energy consumption These methods frequently employ expensive or unstable reagents such as trifluoroacetic acid derivatives that necessitate careful handling and specialized storage facilities thereby increasing overall production costs The substrate scope remains severely restricted due to functional group incompatibilities where sensitive moieties decompose under aggressive reaction environments resulting in low yields typically below acceptable industrial thresholds Moreover conventional approaches demand multi-step pre-activation procedures that introduce additional purification challenges and generate substantial waste streams complicating environmental compliance efforts The narrow tolerance for diverse substituents limits molecular diversity hindering medicinal chemistry optimization efforts required for modern drug discovery programs.
The Novel Approach
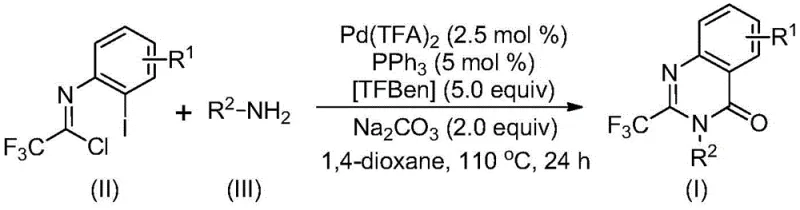
The patented methodology overcomes these constraints through an elegant palladium-catalyzed carbonylation tandem reaction that utilizes cost-effective starting materials including commercially available trifluoroethylimidoyl chloride and amines under mild thermal conditions at precisely controlled temperatures The process eliminates pre-activation requirements by directly coupling substrates through a well-defined catalytic cycle that maintains high efficiency across a broad range of functional groups including halogens alkyl groups and aryl substituents as demonstrated by successful synthesis of fifteen distinct derivatives This innovative approach achieves exceptional yields up to 98% while operating within a practical temperature window of 110°C that minimizes energy consumption and equipment stress The streamlined post-treatment procedure involving simple filtration followed by standard column chromatography significantly reduces processing time compared to conventional multi-step purification protocols The methodology's scalability has been proven from gram-scale laboratory demonstrations to potential commercial production volumes ensuring seamless transition from discovery to manufacturing phases without reoptimization.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with alkali-promoted intermolecular carbon-nitrogen bond coupling between trifluoroethylimidoyl chloride and amine forming a key trifluoroacetamidine intermediate which subsequently undergoes oxidative addition where palladium inserts into the carbon-iodine bond creating a divalent palladium species The critical carbon monoxide equivalent is released from TFBen under thermal activation enabling insertion into the carbon-palladium bond to form an acyl palladium intermediate which then undergoes intramolecular cyclization facilitated by base-promoted palladium-nitrogen bond formation This step generates a seven-membered ring palladium complex that proceeds through reductive elimination to yield the final quinazolinone product while regenerating the active catalyst species The precise stoichiometric balance between palladium trifluoroacetate triphenylphosphine and sodium carbonate at ratios of 0.025:0.05:2 ensures optimal catalyst turnover without decomposition or side reactions The solvent choice of dioxane provides ideal polarity for dissolving both organic substrates and inorganic additives while maintaining catalyst stability throughout the extended reaction period.
Impurity control is achieved through multiple mechanistic safeguards including the selective formation of the desired cyclization product over competing pathways due to the steric and electronic properties of the palladium catalyst system The absence of transition metal residues in final products eliminates costly purification steps required by alternative methods while maintaining stringent purity specifications essential for pharmaceutical applications The reaction's tolerance for various substituents on both aromatic rings prevents unwanted side reactions that typically generate impurities in conventional syntheses The precise control over reaction temperature at exactly 110°C prevents thermal degradation pathways that could lead to decomposition products while ensuring complete conversion within the specified time frame The methodology's inherent selectivity minimizes byproduct formation resulting in cleaner reaction profiles that simplify downstream processing without compromising yield or purity.
How to Synthesize Trifluoromethyl Quinazolinone Efficiently
This patented methodology provides a robust framework for producing high-purity trifluoromethyl quinazolinone intermediates through a carefully optimized catalytic process that eliminates traditional synthetic bottlenecks The procedure leverages commercially available starting materials under mild thermal conditions to achieve exceptional yields while maintaining operational simplicity suitable for both laboratory-scale development and industrial manufacturing environments Detailed standardized synthesis steps including precise reagent quantities temperature control parameters and purification protocols are provided below to ensure consistent results across different production scales This approach represents a significant advancement over conventional methods by removing hazardous reagents and complex multi-step sequences while delivering superior product quality.
- Combine palladium trifluoroacetate (2.5 mol%), triphenylphosphine (5 mol%), TFBen (5.0 equiv), sodium carbonate (2.0 equiv), trifluoroethylimidoyl chloride, and amine in dioxane solvent within a Schlenk tube under inert atmosphere.
- Heat the reaction mixture at precisely 110°C for exactly 24 hours with continuous stirring to ensure complete conversion while maintaining optimal catalyst activity throughout the carbonylation process.
- After reaction completion, filter the mixture through silica gel followed by column chromatography purification using standard elution techniques to isolate the pure trifluoromethyl quinazolinone product with minimal impurities.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional manufacturing approaches The process eliminates dependency on expensive transition metal catalysts requiring complex removal procedures thereby reducing both material costs and processing time while enhancing environmental compliance profiles The use of readily available starting materials ensures consistent supply chain continuity even during market fluctuations while minimizing raw material procurement risks associated with specialized reagents The streamlined manufacturing process significantly reduces production cycle times compared to conventional methods enabling faster response to changing market demands without compromising product quality or regulatory compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activation steps and hazardous reagents substantially lowers raw material costs while simplified purification procedures reduce solvent consumption and waste disposal expenses The process operates under standard temperature conditions avoiding energy-intensive operations required by alternative methods thereby achieving significant operational cost savings without requiring capital investment in specialized equipment.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with broad supplier networks ensures consistent raw material availability while minimizing vulnerability to single-source dependencies The methodology's robustness across diverse production scales provides flexibility to adjust output volumes rapidly in response to changing demand patterns without revalidation requirements.
- Scalability and Environmental Compliance: The seamless transition from laboratory-scale development to commercial production volumes maintains consistent product quality while eliminating toxic metal residues that complicate waste treatment processes The simplified reaction profile generates minimal byproducts reducing environmental impact and facilitating regulatory approval through cleaner manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations regarding this patented methodology based on detailed analysis of its implementation parameters performance metrics and commercial applicability These insights derive directly from experimental data presented in the patent documentation providing authoritative answers to common inquiries from technical procurement teams evaluating this technology for adoption.
Q: What are the key limitations of traditional synthesis methods for trifluoromethyl quinazolinones?
A: Conventional approaches suffer from harsh reaction conditions requiring high temperatures or pressures expensive substrates needing pre-activation steps narrow substrate scope limiting molecular diversity and low yields due to competing side reactions that complicate purification processes significantly hindering industrial scalability.
Q: How does the palladium-catalyzed carbonylation approach improve substrate compatibility and yield?
A: The novel method utilizes readily available starting materials with exceptional functional group tolerance across diverse amine substrates operates under mild conditions without pre-activation achieves consistently high yields up to 98% through optimized catalytic cycles and maintains excellent regioselectivity ensuring broad applicability in complex molecule synthesis.
Q: What specific advantages does this method offer for scaling up pharmaceutical intermediate production?
A: The process demonstrates seamless scalability from gram-scale laboratory batches to multi-kilogram commercial production with straightforward post-treatment procedures maintains stringent purity specifications through simplified purification and eliminates costly metal removal steps resulting in substantial cost savings while ensuring reliable supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Quinazolinone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs that ensure consistent product quality meeting global regulatory standards We specialize in transforming innovative synthetic methodologies like this patented process into reliable manufacturing solutions that deliver exceptional value across pharmaceutical intermediate production Our technical expertise encompasses complex heterocyclic chemistry optimization ensuring seamless integration of novel routes into existing production frameworks while maintaining full compliance with cGMP requirements.
Leverage our technical procurement team's expertise by requesting a Customized Cost-Saving Analysis tailored to your specific manufacturing needs today We provide comprehensive support including specific COA data route feasibility assessments and scalability evaluations to help you optimize your supply chain operations while reducing time-to-market for critical pharmaceutical intermediates.
