Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as the backbone of modern therapeutics. Among these, quinazolinone derivatives stand out due to their profound biological activities, ranging from antifungal and antiviral properties to potent anticancer effects. A significant breakthrough in this domain is detailed in patent CN112480015B, which discloses a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This technology addresses critical bottlenecks in traditional heterocycle synthesis by leveraging a palladium-catalyzed carbonylation cascade that operates under relatively mild conditions. The introduction of the trifluoromethyl group is particularly strategic, as it enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these intermediates invaluable for the development of next-generation active pharmaceutical ingredients (APIs). For R&D directors and procurement specialists, understanding the nuances of this patented methodology offers a pathway to optimizing supply chains and reducing the cost of goods sold for complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that are often fraught with operational hazards and economic inefficiencies. Traditional synthetic routes frequently necessitate the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols, thereby inflating capital expenditure and operational complexity. Furthermore, many established methods depend on ruthenium or platinum catalysts, which are not only prohibitively expensive but also pose significant challenges regarding residual metal removal in final drug substances. Other approaches involve the use of pre-activated substrates such as 2-bromoformylaniline or acid anhydrides, which add extra synthetic steps and generate substantial chemical waste. These conventional processes often suffer from narrow substrate scope, meaning that introducing diverse functional groups requires extensive re-optimization, slowing down the drug discovery timeline. Additionally, low yields and harsh reaction conditions further diminish the commercial viability of these older techniques, creating a pressing need for a more streamlined and cost-effective alternative.
The Novel Approach
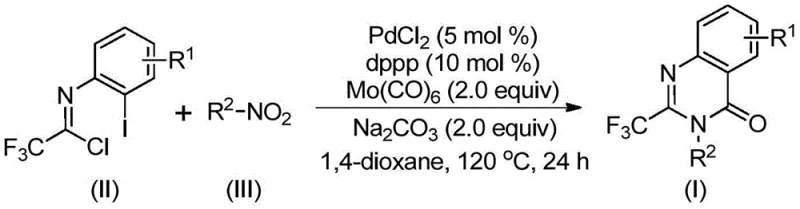
In stark contrast to these legacy methods, the technology described in patent CN112480015B introduces a transformative one-pot strategy that utilizes cheap and readily available nitro compounds as starting materials. This novel approach employs a palladium catalyst system combined with molybdenum hexacarbonyl (Mo(CO)6), which serves as a safe and convenient solid source of carbon monoxide, effectively eliminating the need for high-pressure gas equipment. The reaction proceeds through a tandem sequence where the nitro group is reduced in situ, followed by cyclization to form the quinazolinone ring. This multi-component coupling is highly efficient, demonstrating excellent compatibility with a wide range of functional groups including halogens, alkyls, and trifluoromethyl substituents. By consolidating multiple synthetic transformations into a single vessel, this method drastically reduces solvent consumption, purification time, and overall process mass intensity. The ability to synthesize diverse 2-trifluoromethyl quinazolinone derivatives from simple precursors represents a significant leap forward in process chemistry, offering a scalable solution for industrial manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this synthesis is a sophisticated orchestration of reduction, coupling, and cyclization events driven by the palladium catalyst and the carbonyl source. The process likely initiates with the reduction of the nitro compound to the corresponding amine by Mo(CO)6, which simultaneously releases carbon monoxide upon heating. This generated amine then undergoes a base-promoted intermolecular coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive divalent palladium species. The carbon monoxide released earlier then inserts into this carbon-palladium bond, forming an acyl-palladium intermediate. Under the influence of the base, an intramolecular nucleophilic attack occurs, facilitating the formation of a palladium-nitrogen bond and closing the ring to create a seven-membered cyclic palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. Understanding this intricate cycle is crucial for R&D teams aiming to further optimize reaction parameters or adapt the chemistry to novel substrates.
From an impurity control perspective, the one-pot nature of this reaction offers distinct advantages by minimizing the isolation of unstable intermediates. The use of Mo(CO)6 ensures a steady, controlled release of carbon monoxide, preventing the accumulation of toxic gas and reducing side reactions associated with high CO concentrations. Furthermore, the choice of dioxane as the preferred solvent enhances the solubility of all reagents, promoting homogeneous reaction conditions that lead to higher conversion rates and cleaner crude profiles. The mild basic conditions provided by sodium carbonate are sufficient to drive the cyclization without degrading sensitive functional groups on the aromatic rings. This robustness allows for the synthesis of complex molecules with high purity, reducing the burden on downstream purification processes. For quality assurance teams, the predictable formation of the quinazolinone core with minimal byproduct formation simplifies the validation of the manufacturing process and ensures consistent batch-to-batch quality.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, requiring standard glassware and heating equipment rather than specialized high-pressure reactors. The protocol involves charging a reaction vessel with the palladium catalyst, the dppp ligand, sodium carbonate, Mo(CO)6, the trifluoroethylimidoyl chloride, and the chosen nitro compound in an organic solvent such as 1,4-dioxane. The mixture is then heated to 120°C and stirred for a period ranging from 16 to 30 hours, depending on the specific electronic nature of the substrates. Upon completion, the reaction mixture is cooled, filtered to remove inorganic salts and metal residues, and the filtrate is concentrated. The crude product is typically purified via column chromatography to yield the target compound with high purity. This operational simplicity makes the method highly attractive for rapid scale-up from gram to kilogram quantities.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours under inert atmosphere to facilitate the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver of cost reduction lies in the utilization of nitro compounds as starting materials, which are commodity chemicals available in bulk quantities at a fraction of the cost of pre-functionalized anilines or benzamides required by traditional methods. By eliminating the need for high-pressure carbon monoxide infrastructure, companies can avoid significant capital investments in specialized autoclaves and safety systems, thereby lowering the barrier to entry for manufacturing these valuable intermediates. Furthermore, the one-pot design reduces the number of unit operations, leading to substantial savings in labor, energy, and solvent usage. The high atom economy and reduced waste generation also align with green chemistry principles, potentially lowering waste disposal costs and simplifying environmental compliance.
- Cost Reduction in Manufacturing: The economic advantage of this process is multifaceted, stemming primarily from the substitution of expensive reagents with inexpensive nitroarenes and the avoidance of high-pressure equipment. Traditional methods often require costly noble metal catalysts like ruthenium or platinum, whereas this method utilizes a more economical palladium system with a ligand that can potentially be recovered or used in lower loadings. The elimination of pre-activation steps for the starting materials further trims the synthetic timeline, reducing man-hours and utility consumption per kilogram of product. Additionally, the high yields reported across a broad substrate scope mean that less raw material is wasted on failed batches or difficult purifications, directly improving the overall process mass intensity and cost efficiency.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available commodity chemicals rather than bespoke, custom-synthesized building blocks. Nitro compounds and trifluoroethylimidoyl chlorides are commercially accessible from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a variety of functional groups, allows for flexibility in sourcing; if a specific substituted nitro compound is unavailable, alternative analogs can often be substituted without requiring a complete re-development of the process. This flexibility ensures continuity of supply even in volatile market conditions, providing procurement teams with greater leverage in negotiations and inventory management.
- Scalability and Environmental Compliance: Scaling this process from the bench to commercial production is facilitated by the absence of hazardous high-pressure gases and the use of standard heating protocols. The use of Mo(CO)6 as a solid CO surrogate simplifies reactor design and operation, making it easier to transfer the technology to large-scale stainless steel reactors without extensive engineering modifications. From an environmental standpoint, the reduced solvent usage and lower waste generation inherent in the one-pot design contribute to a smaller environmental footprint. This aligns with increasingly stringent global regulations on chemical manufacturing emissions and waste, ensuring long-term operational sustainability and reducing the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical application of this technology for pharmaceutical and fine chemical manufacturing. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing production portfolios.
Q: What are the advantages of using nitro compounds over traditional anilines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-functionalized anilines or benzamides. This method utilizes them directly, avoiding costly pre-activation steps and reducing overall raw material expenses while maintaining high reaction efficiency.
Q: Does this method require high-pressure carbon monoxide equipment?
A: No, this novel approach utilizes Mo(CO)6 as a solid carbon monoxide substitute. This eliminates the need for dangerous high-pressure CO gas cylinders and specialized autoclaves, significantly enhancing operational safety and simplifying the reactor requirements for scale-up.
Q: What is the substrate scope for the R1 and R2 groups?
A: The method demonstrates excellent functional group tolerance. R1 can be hydrogen, alkyl, halogens, or trifluoromethyl groups, while R2 accommodates alkyl, cycloalkyl, and various substituted aryl groups, allowing for the synthesis of diverse libraries for SAR studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and securing supply chains. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. We are committed to delivering high-quality 2-trifluoromethyl quinazolinone derivatives that adhere to the highest standards of safety and efficacy, leveraging our deep expertise in palladium-catalyzed transformations to optimize yield and minimize impurities.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply of complex heterocyclic intermediates, empowering your R&D efforts and ensuring the commercial success of your therapeutic candidates.
