Advanced Catalytic Synthesis of Benzofuran-2(3H)-one for Scalable Agrochemical and Pharmaceutical Production
Introduction to the Novel Synthetic Route
The global demand for high-performance agrochemicals and pharmaceutical intermediates continues to drive innovation in organic synthesis, particularly for core heterocyclic structures like benzofuran-2(3H)-one. As detailed in the recent patent CN114478448A, a groundbreaking method has been developed that redefines the efficiency of producing this critical scaffold. This molecule serves as a pivotal building block for the synthesis of azoxystrobin, a widely used strobilurin fungicide, as well as various novel antioxidants and pharmaceutical agents. The disclosed technology addresses long-standing inefficiencies in traditional manufacturing by streamlining the reaction pathway from readily available starting materials. By leveraging advanced phase transfer catalysis and high-pressure hydrolysis techniques, this new methodology offers a robust alternative that promises to enhance both the economic viability and the environmental profile of large-scale production. For procurement leaders and R&D directors seeking a reliable agrochemical intermediate supplier, understanding the nuances of this patented route is essential for securing a competitive supply chain.
The significance of this invention lies not merely in the chemical transformation itself, but in the holistic optimization of the process parameters. Traditional routes often suffer from multi-step complexities, hazardous reagent handling, and poor atom economy, which collectively inflate the cost of goods sold (COGS). In contrast, the approach outlined in patent CN114478448A demonstrates a meticulous balance between reaction kinetics and operational simplicity. The process initiates with the cyanation of o-chlorobenzyl chloride, proceeds through a controlled saponification and high-pressure hydrolysis, and culminates in an elegant acid-catalyzed cyclization. Each stage has been engineered to maximize yield while minimizing waste generation, aligning perfectly with modern green chemistry principles. This report provides a deep technical dissection of this pathway, offering actionable insights for stakeholders focused on cost reduction in pharmaceutical intermediates manufacturing and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzofuran-2(3H)-one and its derivatives has been plagued by convoluted reaction sequences that introduce significant operational bottlenecks. Conventional methodologies frequently rely on harsh reaction conditions that necessitate specialized corrosion-resistant equipment, thereby driving up capital expenditure. Furthermore, older protocols often involve the use of stoichiometric amounts of expensive or toxic reagents, which complicates downstream purification and waste disposal. For instance, traditional cyclization strategies might require prolonged heating times or the use of strong dehydrating agents that generate substantial acidic waste streams. These factors contribute to a fragmented production workflow where yield losses accumulate at each stage, ultimately eroding profit margins. Additionally, the lack of effective catalytic systems in legacy processes often results in incomplete conversions, leading to complex impurity profiles that are difficult to separate. This variability poses a severe risk to supply chain consistency, making it challenging for manufacturers to guarantee the high-purity OLED material or agrochemical standards required by downstream clients.
The Novel Approach
The innovative strategy presented in patent CN114478448A effectively dismantles these barriers through a rational redesign of the synthetic logic. By selecting o-chlorobenzyl chloride as the primary feedstock, the process capitalizes on the availability and low cost of commodity chemicals. The introduction of a phase transfer catalyst in the initial cyanation step dramatically accelerates the reaction rate under mild conditions, eliminating the need for extreme temperatures or pressures at the outset. Subsequently, the integration of a high-pressure hydrolysis step allows for the direct conversion of the nitrile intermediate into the requisite hydroxy-acid precursor with exceptional efficiency. This telescoped approach reduces the total number of isolation steps, thereby minimizing material handling and potential product loss. The final cyclization is achieved using a catalytic amount of acid in a recyclable solvent system, ensuring that the process remains economically sustainable. This streamlined workflow represents a paradigm shift, offering a clear pathway for the commercial scale-up of complex polymer additives and fine chemical intermediates with superior reproducibility.
Mechanistic Insights into the Catalytic Cascade
The success of this synthesis hinges on the precise orchestration of four distinct chemical transformations, each optimized for maximum throughput. The journey begins with the nucleophilic substitution of o-chlorobenzyl chloride (OCBC) by sodium cyanide. In this critical first step, the presence of a phase transfer catalyst, such as benzyltriethylammonium chloride, facilitates the transport of the cyanide anion into the organic phase, overcoming the solubility limitations inherent in biphasic systems. This interaction occurs efficiently at 80°C, yielding o-chlorophenylacetonitrile (OCBN) with reported yields reaching 95%. The mechanistic elegance here lies in the catalyst's ability to lower the activation energy of the substitution, allowing the reaction to proceed rapidly without the formation of significant byproducts.
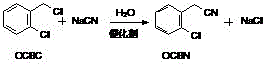
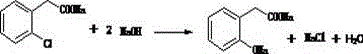
Following the formation of the nitrile, the process advances to a saponification stage where OCBN is treated with sodium hydroxide at reflux temperatures around 110°C. This step converts the nitrile group into a carboxylate salt, specifically sodium o-chlorophenylacetate (OCPANa), while releasing ammonia gas which is safely scrubbed. The true technological breakthrough, however, is observed in the subsequent high-pressure hydrolysis. Here, the chloro-substituted acetate undergoes a nucleophilic aromatic substitution under vigorous conditions (220°C, 1.5-2.1 MPa) in the presence of a copper-based catalyst. This transformative step replaces the chlorine atom with a hydroxyl group, generating the disodium salt of o-hydroxyphenylacetic acid. The use of copper catalysis is pivotal, as it activates the aromatic ring towards nucleophilic attack, a transformation that would otherwise require prohibitively harsh conditions.
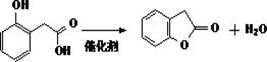
The final stage involves the acidification of the disodium salt to liberate o-hydroxyphenylacetic acid (OHPAA), followed by an intramolecular esterification to close the lactone ring. This cyclization is catalyzed by strong acids such as sulfuric acid or p-toluenesulfonic acid in a solvent like toluene. The reaction utilizes azeotropic distillation to continuously remove water, driving the equilibrium towards the formation of benzofuran-2(3H)-one. The thermodynamic favorability of forming the five-membered lactone ring, combined with the efficient removal of the condensation byproduct, ensures high conversion rates. This mechanistic sequence not only guarantees high purity but also simplifies the isolation of the final product through standard distillation techniques, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Benzofuran-2(3H)-one Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to ensure safety and optimal yield. The process is divided into four logical stages: cyanation, saponification, high-pressure hydrolysis, and cyclization. Each step builds upon the previous one, creating a continuous flow of value-added intermediates. Operators must pay particular attention to the temperature controls during the exothermic cyanation and the pressure management during the hydrolysis phase. Detailed standard operating procedures (SOPs) are essential to manage the handling of cyanide salts and high-pressure reactors safely. For a comprehensive guide on the exact molar ratios, stirring speeds, and workup protocols, please refer to the standardized synthesis steps provided below.
- React o-chlorobenzyl chloride with sodium cyanide using a phase transfer catalyst at 80°C to form o-chlorophenylacetonitrile (OCBN).
- Hydrolyze OCBN with sodium hydroxide at reflux (110°C) to produce solid sodium o-chlorophenylacetate (OCPANa).
- Subject OCPANa to high-pressure alkaline hydrolysis at 220°C with a copper catalyst to form the disodium salt, followed by acidification.
- Perform intramolecular cyclization of o-hydroxyphenylacetic acid in toluene with an acid catalyst to yield benzofuran-2(3H)-one.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages that extend beyond simple chemical yield. For procurement managers tasked with optimizing the budget for agrochemical intermediate sourcing, the elimination of expensive transition metal catalysts in the final steps and the use of commodity reagents translate directly into substantial cost savings. The streamlined nature of the process reduces the consumption of solvents and energy, as fewer distillation and purification cycles are required compared to legacy methods. Furthermore, the high selectivity of the reactions minimizes the generation of hazardous waste, lowering the costs associated with environmental compliance and waste disposal. These factors combine to create a highly competitive cost structure that can withstand market volatility.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing inexpensive starting materials like o-chlorobenzyl chloride and sodium cyanide, which are globally available in bulk quantities. The high yields reported in the patent examples, such as 95% in the initial cyanation and 100% in the saponification step, indicate minimal raw material wastage. By avoiding the use of precious metal catalysts in the final cyclization step and opting for recoverable acid catalysts, the operational expenditure is drastically reduced. Additionally, the simplified workup procedures, which rely on phase separation and crystallization rather than complex chromatography, further decrease the labor and utility costs associated with production.
- Enhanced Supply Chain Reliability: One of the most compelling benefits of this route is its reliance on a robust and stable supply of raw materials. Unlike synthetic pathways that depend on niche or geographically constrained reagents, the inputs for this process are standard industrial chemicals with established supply chains. This reduces the risk of production stoppages due to raw material shortages. Moreover, the process is designed to be scalable, meaning that suppliers can easily ramp up production volumes to meet surging demand without requiring entirely new infrastructure. This reliability is crucial for maintaining the continuity of supply for critical downstream products like fungicides and pharmaceuticals.
- Scalability and Environmental Compliance: The engineering design of this synthesis facilitates easy scale-up from laboratory to industrial plant. The use of standard reactors for the initial steps and conventional autoclaves for the high-pressure step ensures that the technology can be deployed in existing manufacturing facilities. From an environmental standpoint, the process generates fewer byproducts and utilizes solvent recovery systems, such as the recycling of toluene in the final step. The treatment of cyanide-containing wastewater is explicitly addressed in the protocol, ensuring that the process meets stringent environmental regulations. This commitment to sustainability enhances the corporate social responsibility profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of benzofuran-2(3H)-one using this advanced catalytic route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the feasibility, safety, and quality aspects of the process for potential partners and technical evaluators.
Q: What are the key advantages of this synthesis route over traditional methods?
A: This route significantly shortens the synthesis steps compared to conventional processes. It utilizes readily available raw materials like o-chlorobenzyl chloride and employs efficient phase transfer catalysis, which simplifies the workup procedure and reduces overall production costs.
Q: How is the quality and purity of the final benzofuran-2(3H)-one ensured?
A: The process includes rigorous purification steps such as distillation under vacuum and multiple washing stages (alkali and water washes). The use of specific catalysts like benzyltriethylammonium chloride and copper acetate ensures high conversion rates and minimizes side reactions, leading to a high-purity final product suitable for sensitive agrochemical applications.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is designed for industrial scalability. While step 3 requires high-pressure equipment (autoclave), the conditions (220°C, 1.5-2.1 MPa) are well within standard industrial capabilities. The subsequent steps utilize common solvents like toluene and standard distillation techniques, facilitating easy scale-up from pilot to commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran-2(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates requires more than just a patent; it demands engineering excellence and unwavering quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to full-scale manufacturing is seamless. We are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications required by the global agrochemical and pharmaceutical industries. Our commitment to technical precision ensures that every batch of benzofuran-2(3H)-one delivered meets the highest standards of consistency and performance.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
