Advanced Metal-Free Synthesis Strategy for Scalable Production of Trifluoromethylated Triazole Intermediates
Advanced Metal-Free Synthesis Strategy for Scalable Production of Trifluoromethylated Triazole Intermediates
The pharmaceutical industry continuously seeks robust and economically viable pathways for constructing complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN113105402B introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value active pharmaceutical ingredients (APIs) such as Sitagliptin, Maraviroc, and Deferasirox. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by leveraging a metal-free, iodine-promoted cascade reaction that operates under remarkably mild and operationally simple conditions. By utilizing dimethyl sulfoxide (DMSO) as both solvent and oxidant participant, this technology enables the direct assembly of trifluoromethylated triazoles from inexpensive aryl ethyl ketones and trifluoroethylimide hydrazides. For R&D directors and procurement strategists, this represents a paradigm shift towards greener, more cost-effective manufacturing processes that maintain high purity standards while drastically simplifying the supply chain for essential pharmaceutical intermediates.
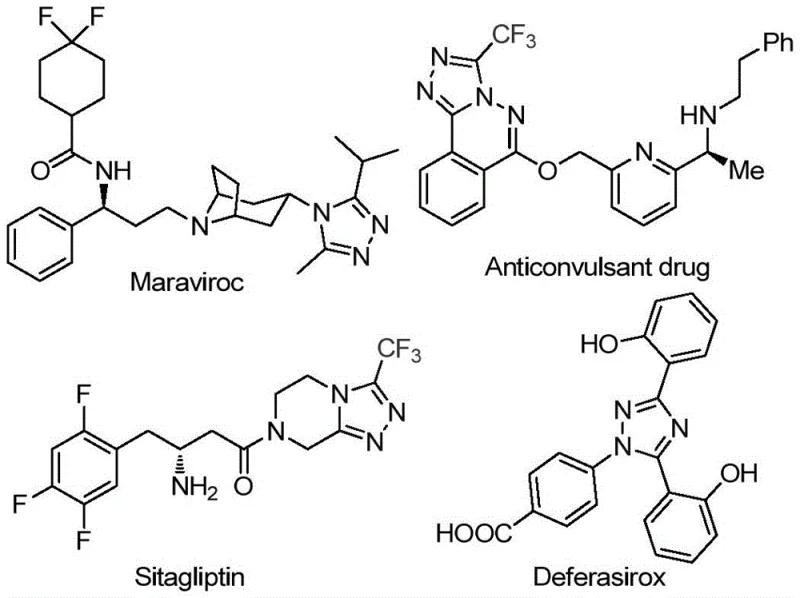
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, particularly those bearing both trifluoromethyl and acyl groups, has been plagued by significant synthetic challenges that hinder efficient commercial scale-up. Conventional methodologies often rely heavily on the use of toxic and expensive transition metal catalysts, such as copper or palladium complexes, which necessitate rigorous anhydrous and oxygen-free reaction environments to prevent catalyst deactivation. Furthermore, the presence of residual heavy metals in the final product poses a severe regulatory hurdle for pharmaceutical applications, requiring extensive and costly purification steps involving specialized scavengers or repeated recrystallizations to meet stringent ICH Q3D guidelines. These traditional routes frequently suffer from limited substrate scope, poor functional group tolerance, and low atom economy, resulting in substantial waste generation and inflated production costs that ultimately impact the profitability of the final drug product.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a non-metallic iodine promotion system that fundamentally simplifies the synthetic workflow while expanding the accessible chemical space. This methodology capitalizes on the unique reactivity of elemental iodine in conjunction with DMSO to facilitate a tandem iodination and Kornblum oxidation sequence, generating reactive aryl diketone intermediates in situ. These intermediates then undergo condensation with trifluoroethylimide hydrazides followed by oxidative cyclization to yield the desired 3,4,5-trisubstituted 1,2,4-triazoles with high efficiency. The process is exceptionally robust, tolerating a wide array of substituents on both the aryl ketone and the hydrazide components, including electron-donating and electron-withdrawing groups, as evidenced by the successful synthesis of diverse derivatives shown in the structural examples. This operational simplicity eliminates the need for inert atmosphere techniques, making the process highly amenable to standard reactor setups found in most multipurpose chemical manufacturing facilities.
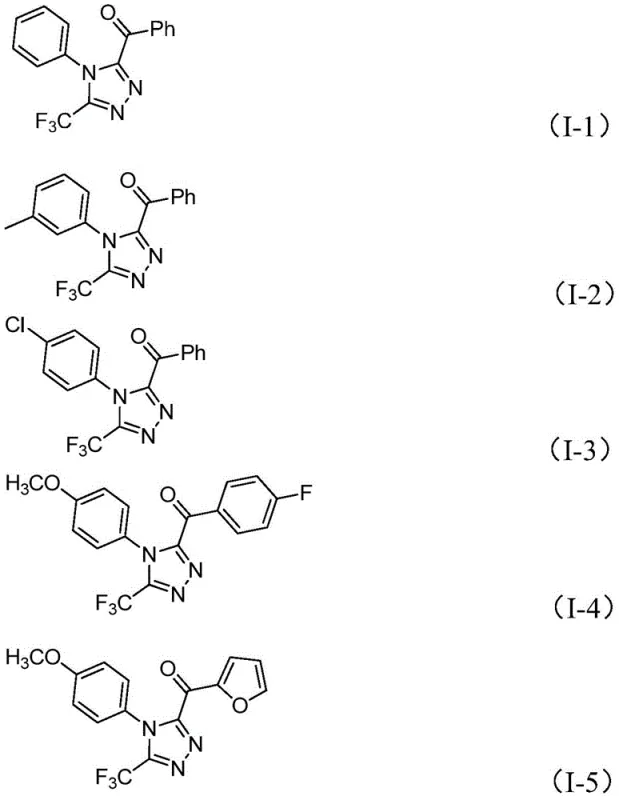
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation events driven by the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes alpha-iodination followed by Kornblum oxidation, where DMSO acts as the oxygen source to convert the methyl ketone into an alpha-dicarbonyl species. This highly electrophilic intermediate is then trapped by the nucleophilic nitrogen of the trifluoroethylimide hydrazide, forming a hydrazone linkage through a dehydration process. Subsequent intramolecular cyclization, promoted by the basic environment provided by pyridine and sodium dihydrogen phosphate alongside the oxidizing power of excess iodine, closes the triazole ring. This cascade effectively installs the trifluoromethyl group at the 3-position and the acyl group at the 5-position in a single pot, avoiding the isolation of unstable intermediates. The precise control over reaction temperature, typically ramping from 90-110°C to 110-130°C, ensures optimal kinetics for each step of the cascade, minimizing side reactions and maximizing the yield of the target heterocycle.
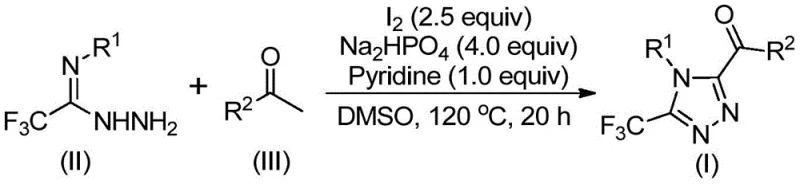
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of metal-complexed byproducts that are notoriously difficult to separate. The primary byproducts are inorganic salts and reduced iodine species, which are water-soluble and easily removed during the aqueous workup phase. The use of pyridine and phosphate buffers helps maintain a stable pH environment that suppresses the hydrolysis of the sensitive hydrazone intermediate, thereby enhancing the overall mass balance of the reaction. For quality control teams, this translates to a cleaner crude profile that requires less aggressive chromatographic purification, reducing solvent consumption and processing time. The ability to fine-tune the electronic properties of the starting materials allows chemists to predictably modulate the reactivity, ensuring consistent batch-to-batch reproducibility which is critical for GMP manufacturing environments.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The execution of this synthesis protocol is designed for maximum operational ease, allowing chemists to achieve high yields without specialized equipment or extreme safety measures. The process begins with the dissolution of the aryl ethyl ketone and a stoichiometric amount of iodine in DMSO, followed by a controlled heating period to generate the oxidized intermediate. Once this initial transformation is complete, the remaining reagents, including the hydrazide, base, and additional iodine, are introduced directly into the reaction vessel, streamlining the workflow into a concise one-pot procedure. Detailed standardized synthesis steps for implementing this protocol in a pilot or production setting are provided below to ensure technical accuracy and safety compliance.
- Mix aryl ethyl ketone and elemental iodine in DMSO and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits that directly enhance the bottom line. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth elements or noble metals. Furthermore, the reliance on commodity chemicals like aryl ketones and elemental iodine ensures a stable and diversified supply base, reducing the vulnerability to single-source supplier disruptions. The simplified downstream processing, characterized by the absence of heavy metal scavenging steps, leads to substantial reductions in auxiliary material costs and waste disposal fees, contributing to a more sustainable and cost-efficient manufacturing lifecycle.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive elemental iodine results in a drastic decrease in raw material expenditure per kilogram of product. Additionally, the avoidance of complex metal removal protocols significantly shortens the production cycle time and reduces the consumption of high-purity solvents and specialized filtration media. This streamlined process flow lowers the overall cost of goods sold (COGS), providing a competitive pricing advantage in the global market for pharmaceutical intermediates while maintaining high profit margins.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as substituted acetophenones and commercially sourced iodine minimizes lead times and ensures consistent availability even during periods of market volatility. The robustness of the reaction conditions, which do not require stringent exclusion of moisture or oxygen, reduces the risk of batch failures due to environmental factors, thereby guaranteeing reliable delivery schedules to downstream API manufacturers. This stability is crucial for maintaining continuous production lines and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The protocol has been validated for scalability, demonstrating seamless translation from gram-scale laboratory experiments to multi-kilogram production batches without loss of efficiency or selectivity. The absence of toxic heavy metals simplifies the environmental permitting process and reduces the burden of hazardous waste management, aligning with increasingly strict global environmental regulations. This green chemistry approach not only enhances the corporate sustainability profile but also future-proofs the manufacturing asset against tightening regulatory constraints on metal residues in drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on feasibility and performance metrics. Understanding these aspects is vital for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metallic promoter, completely eliminating the need for costly heavy metal catalysts and the subsequent complex removal steps required for pharmaceutical grade purity.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) under aerobic conditions, involving a two-stage heating process initially at 90-110°C and subsequently at 110-130°C, without the need for strict anhydrous or oxygen-free environments.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability, utilizing cheap and readily available starting materials like aryl ethyl ketones, and has been demonstrated to expand easily from gram-level laboratory synthesis to commercial scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to leverage this advanced synthesis technology for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from process development to full-scale manufacturing is seamless and efficient. We operate state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest quality standards required for pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how this metal-free route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume requirements. We encourage potential partners to contact us immediately to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next generation of triazole-based therapeutics reaches the market faster and more economically.
