Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Applications
Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN113105402B introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, specifically targeting those bearing both trifluoromethyl and acyl groups. This innovation addresses a significant gap in organic synthesis, where introducing a trifluoromethyl group into a heterocyclic molecule is known to drastically enhance physicochemical properties such as metabolic stability, lipophilicity, and bioavailability. The disclosed methodology leverages a non-metallic iodine-promoted system, departing from traditional reliance on precious metal catalysts. By utilizing cheap and readily available aryl ethanones and trifluoroethylimine hydrazides, this process offers a streamlined pathway to complex molecular architectures found in high-value drugs like Sitagliptin and Deferasirox.
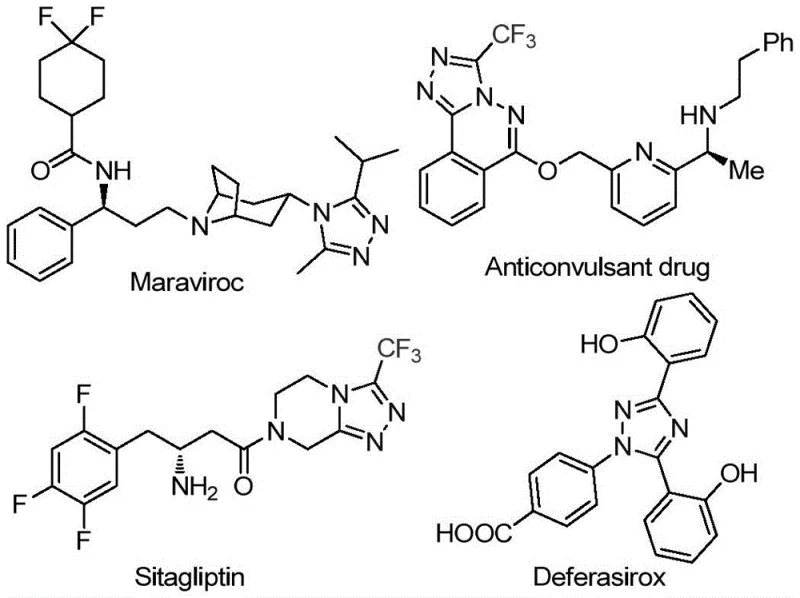
The strategic importance of this technology cannot be overstated for R&D directors aiming to optimize impurity profiles and reduce raw material costs. The patent details a tandem reaction sequence involving iodination and Kornblum oxidation, followed by intramolecular cyclization. This approach not only simplifies the operational workflow by avoiding stringent anhydrous and oxygen-free conditions but also ensures high functional group tolerance. For procurement and supply chain stakeholders, the shift towards commodity chemicals like elemental iodine and dimethyl sulfoxide represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. The ability to scale this reaction from laboratory benchtop to industrial tonnage without compromising yield or purity positions this technology as a cornerstone for reliable pharmaceutical intermediate supplier networks seeking sustainable and efficient production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, particularly those incorporating electron-withdrawing trifluoromethyl groups, has been plagued by significant technical and economic hurdles. Traditional methodologies often rely heavily on transition metal catalysts such as copper, palladium, or rhodium complexes to facilitate C-N bond formation and ring closure. These heavy metal catalysts are not only prohibitively expensive but also introduce severe downstream processing challenges, including the necessity for rigorous metal scavenging steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients (APIs). Furthermore, many conventional routes require harsh reaction conditions, including extreme temperatures, strong bases, or strictly anhydrous environments, which increase energy consumption and operational complexity. The limited substrate scope of these older methods often restricts the diversity of substituents that can be introduced at the 3, 4, and 5 positions, thereby limiting the chemical space available for medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN113105402B revolutionizes this landscape by employing a metal-free, iodine-promoted strategy that is both economically and environmentally superior. This method utilizes elemental iodine, a cheap and abundant halogen, acting as both an oxidant and a promoter in conjunction with dimethyl sulfoxide (DMSO). The process initiates with the iodination and subsequent Kornblum oxidation of aryl ethanones to generate reactive aryl diketone intermediates in situ. These intermediates then undergo condensation with trifluoroethylimine hydrazides, followed by an iodine-mediated intramolecular cyclization to forge the triazole ring. This cascade reaction eliminates the need for pre-functionalized substrates or exotic reagents. The operational simplicity is further enhanced by the tolerance to moisture and oxygen, allowing reactions to be conducted under ambient atmospheric conditions. This robustness significantly lowers the barrier to entry for commercial scale-up, making it an ideal candidate for high-purity pharmaceutical intermediate production where consistency and cost-efficiency are paramount.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation chemistry driven by the unique redox properties of the iodine-DMSO system. Initially, the aryl ethanone substrate reacts with molecular iodine in DMSO to form an alpha-iodo ketone intermediate. This species is highly susceptible to nucleophilic attack by DMSO, leading to the formation of a sulfonium salt which, upon hydrolysis or elimination, yields the corresponding aryl glyoxal or 1,2-diketone derivative. This Kornblum-type oxidation is the critical activation step that generates the electrophilic center necessary for the subsequent heterocycle formation. Once the dicarbonyl species is generated, it reacts with the nucleophilic nitrogen of the trifluoroethylimine hydrazide to form a hydrazone intermediate through a dehydration condensation process. This step is facilitated by the basic environment provided by additives such as sodium dihydrogen phosphate and pyridine, which help neutralize the hydrogen iodide byproduct and drive the equilibrium forward.
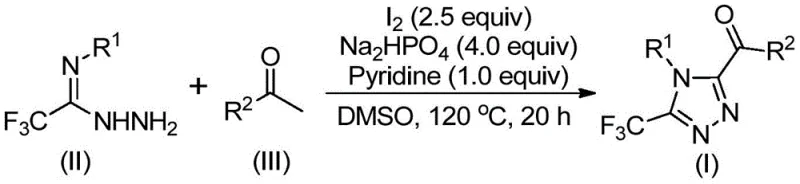
Following hydrazone formation, the reaction enters the cyclization phase, which is the defining step for constructing the 1,2,4-triazole core. Under the continued influence of iodine and elevated temperatures (110-130°C), the hydrazone undergoes an intramolecular nucleophilic attack where the terminal nitrogen attacks the adjacent carbonyl carbon or the activated imine carbon, depending on the specific tautomeric state. The presence of iodine likely facilitates this by activating the carbonyl group or stabilizing the transition state through halogen bonding interactions. The final aromatization step releases water and potentially HI, resulting in the stable, fully conjugated 3,4,5-trisubstituted 1,2,4-triazole system. Understanding this mechanism is crucial for impurity control, as side reactions such as over-oxidation of the ketone or polymerization of the hydrazide can be minimized by precisely controlling the stoichiometry of iodine (typically 2.5 equivalents) and the reaction temperature profile. This deep mechanistic understanding allows process chemists to fine-tune parameters for maximum yield and minimal byproduct formation.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis relies on a carefully orchestrated two-stage heating protocol within a polar aprotic solvent system. To achieve optimal results, practitioners must adhere to specific molar ratios and temperature gradients that maximize the conversion of starting materials while minimizing decomposition. The process begins with the dissolution of the aryl ethanone and a portion of the iodine in DMSO, followed by an initial heating period to generate the oxidative intermediate. Subsequently, the remaining reagents, including the hydrazide, base, and additional iodine, are introduced to trigger the cyclization. Detailed standardized operating procedures regarding addition rates, stirring speeds, and workup protocols are essential for reproducibility. For a comprehensive guide on the exact experimental steps, reagents quantities, and purification techniques required to replicate this high-yielding transformation, please refer to the structured synthesis guide below.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route offers transformative advantages that directly impact the bottom line and operational resilience. The primary driver of value is the drastic simplification of the raw material portfolio. By replacing expensive transition metal catalysts and specialized ligands with commodity chemicals like elemental iodine, sodium salts, and pyridine, manufacturers can significantly reduce the direct material costs associated with production. Furthermore, the elimination of heavy metals removes the need for complex and costly purification steps such as chromatography on metal-scavenging resins or repeated recrystallizations designed to lower metal content below ppm levels. This streamlining of the downstream processing workflow translates into shorter cycle times and higher throughput, effectively increasing the capacity of existing manufacturing facilities without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from both input costs and processing efficiency. Since the catalyst system relies on inexpensive iodine rather than precious metals like palladium or platinum, the volatility of noble metal markets no longer poses a risk to production budgets. Additionally, the reaction does not require specialized anhydrous solvents or inert gas atmospheres (nitrogen or argon), which reduces utility costs and simplifies reactor setup. The high atom economy of the tandem reaction means less waste generation per kilogram of product, lowering waste disposal fees. Consequently, the overall cost of goods sold (COGS) for these valuable triazole intermediates can be substantially optimized, providing a competitive pricing advantage in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the widespread availability of the key reagents. Aryl ethanones and trifluoroethylimine hydrazides are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate ambient moisture and oxygen, means that production is less susceptible to delays caused by strict environmental controls or equipment failures related to gloveboxes or drying columns. This reliability ensures consistent delivery schedules for downstream API manufacturers. Moreover, the scalability of the process from gram to multi-kilogram scales allows for flexible production planning, enabling suppliers to respond rapidly to fluctuating market demands without lengthy re-validation periods typically associated with changing synthetic routes.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The avoidance of toxic heavy metals reduces the environmental footprint of the manufacturing process and simplifies regulatory compliance regarding hazardous waste discharge. DMSO, while requiring careful handling, is a widely accepted solvent with established recovery and recycling protocols. The simplified workup procedure, often involving filtration and standard column chromatography or crystallization, minimizes the volume of organic solvents used compared to multi-step metal-catalyzed sequences. This ease of scale-up ensures that the transition from pilot plant to commercial production is smooth and predictable, mitigating the technical risks often encountered when scaling complex heterocyclic syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, operational parameters, and potential applications. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The answers reflect the specific advantages of the iodine-promoted system over traditional methods, highlighting its versatility and efficiency.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, eliminating the need for costly and toxic heavy metal catalysts typically found in traditional triazole synthesis.
Q: What are the optimal reaction conditions for this cyclization?
A: The reaction proceeds efficiently in dimethyl sulfoxide (DMSO) solvent. It involves a two-stage heating process: initial oxidation at 90-110°C followed by cyclization at 110-130°C, without requiring strict anhydrous or oxygen-free environments.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily scalable to gram levels and beyond. The use of cheap, commercially available starting materials and simple post-processing makes it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the potential of the iodine-promoted synthesis described in patent CN113105402B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to unlock the full potential of this efficient synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our expertise in metal-free catalysis and process optimization can drive value and efficiency in your supply chain, ensuring a reliable source of high-quality intermediates for your drug development programs.
