Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways for constructing complex heterocyclic scaffolds that serve as the backbone of modern therapeutics. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value drugs such as Maraviroc, Sitagliptin, and Deferasirox. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by introducing a metal-free, iodine-promoted protocol that operates under remarkably mild conditions. By leveraging dimethyl sulfoxide (DMSO) as both solvent and oxidant, this technology enables the direct assembly of trifluoromethyl-functionalized triazoles from readily available aryl ethyl ketones. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply of high-purity pharmaceutical intermediates while mitigating the risks associated with heavy metal contamination.
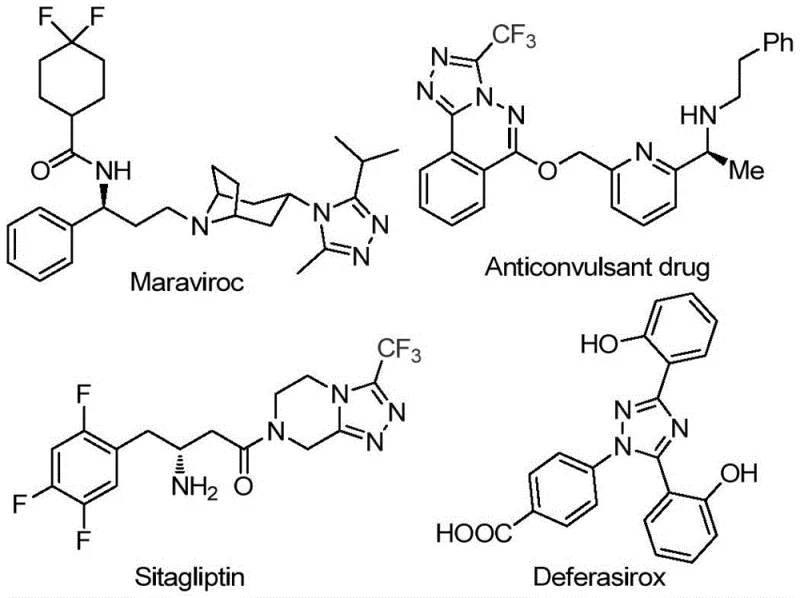
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings has relied heavily on transition metal catalysis or harsh cyclization conditions that pose significant challenges for industrial application. Traditional routes often necessitate the use of expensive palladium or copper catalysts, which not only inflate the raw material costs but also introduce stringent purification requirements to meet residual metal specifications for active pharmaceutical ingredients (APIs). Furthermore, many existing protocols demand strictly anhydrous and oxygen-free environments, requiring specialized equipment and inert gas handling that complicate process safety and increase operational expenditures. The inability to tolerate diverse functional groups without protecting group strategies further limits the versatility of these conventional methods, often resulting in lower overall yields and extended production timelines that hinder rapid scale-up efforts.
The Novel Approach
In stark contrast, the methodology outlined in CN113105402B utilizes a simple yet powerful iodine-promoted system that eliminates the need for transition metals entirely. This approach capitalizes on the dual role of iodine and DMSO to facilitate a tandem oxidation-cyclization sequence, transforming simple aryl ethyl ketones into complex triazole architectures with high efficiency. The process tolerates a wide range of substituents, including halogens, alkoxy groups, and trifluoromethyl moieties, allowing for the rapid generation of diverse chemical libraries as exemplified by compounds I-1 through I-5. By operating without the need for rigorous exclusion of air or moisture, this novel route drastically simplifies the operational workflow, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to reduce lead times and enhance production flexibility.
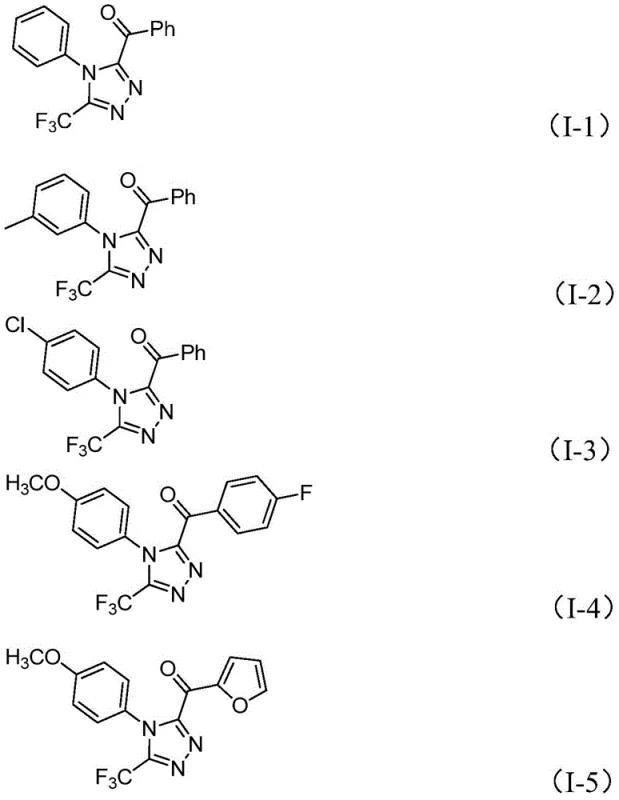
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this synthetic breakthrough lies in a sophisticated cascade reaction mechanism initiated by the iodination and subsequent Kornblum oxidation of the aryl ethyl ketone substrate. In the presence of DMSO, elemental iodine activates the methyl group of the ketone, leading to the formation of an aryl diketone intermediate through an oxidative pathway that avoids the use of stoichiometric oxidants. This reactive diketone species then undergoes a condensation reaction with trifluoroethylimide hydrazide, forming a hydrazone intermediate that serves as the precursor for ring closure. The precise control of reaction temperature and the stoichiometric balance of sodium dihydrogen phosphate and pyridine are critical in driving this equilibrium towards the desired hydrazone without premature degradation of the sensitive trifluoromethyl group.
Following the condensation step, the system undergoes an intramolecular cyclization promoted by the continued presence of iodine and base,最终 yielding the stable 3,4,5-trisubstituted 1,2,4-triazole core. This mechanism is particularly advantageous because it integrates the installation of the acyl group and the trifluoromethyl moiety in a single pot, minimizing unit operations and solvent waste. The tolerance of the reaction to various electronic environments on the aromatic rings suggests a robust radical or electrophilic pathway that is less sensitive to steric hindrance than metal-catalyzed cross-couplings. For process chemists, understanding this mechanism allows for fine-tuning of reaction parameters to maximize yield and minimize impurity profiles, ensuring the delivery of high-purity OLED material or pharmaceutical precursors that meet rigorous quality standards.
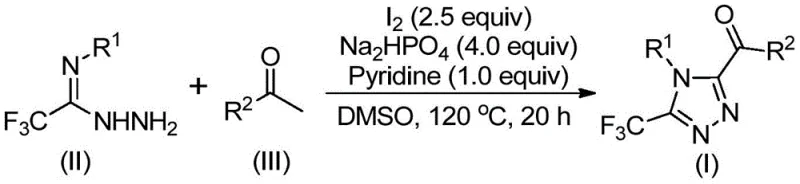
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The implementation of this synthesis route is designed for straightforward execution in standard chemical processing facilities, requiring only common reagents and basic heating equipment. The protocol begins with the dissolution of aryl ethyl ketone and iodine in DMSO, followed by a controlled heating phase to generate the oxidative intermediate. Subsequent addition of the hydrazide component and buffering agents initiates the cyclization, which proceeds to completion over a defined period at elevated temperatures. Detailed standardized synthesis steps see the guide below for precise molar ratios and temperature ramps optimized for maximum conversion.
- Oxidation Phase: React aryl ethyl ketone with elemental iodine in DMSO at 90-110°C for 4-6 hours to generate the aryl diketone intermediate via Kornblum oxidation.
- Condensation Phase: Add trifluoroethylimide hydrazide, additional iodine, sodium dihydrogen phosphate, and pyridine to the mixture.
- Cyclization and Workup: Heat the reaction to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this iodine-promoted methodology offers substantial strategic advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. By removing the dependency on precious metal catalysts, the process inherently lowers the bill of materials and eliminates the costly downstream processing steps typically required for metal scavenging and removal. This simplification translates directly into improved margin potential and a more resilient supply chain that is less vulnerable to fluctuations in the prices of rare earth elements or transition metals. Additionally, the use of commodity chemicals like DMSO and elemental iodine ensures that raw material sourcing remains stable and predictable, reducing the risk of production stoppages due to supply shortages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper significantly decreases the direct material costs associated with each batch production cycle. Furthermore, the absence of heavy metals removes the necessity for specialized purification resins or extensive washing protocols, thereby reducing solvent consumption and waste disposal expenses. This streamlined workflow allows for a more efficient allocation of resources, enabling manufacturers to achieve substantial cost savings while maintaining high product quality standards required for regulated industries.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like aryl ethyl ketones and trifluoroethylimide hydrazides ensures a consistent and reliable supply of inputs for continuous manufacturing operations. The robustness of the reaction conditions, which do not require stringent anhydrous or oxygen-free environments, minimizes the risk of batch failures due to environmental factors or equipment leaks. This operational stability enhances the overall reliability of the supply chain, allowing partners to confidently plan inventory levels and meet delivery commitments without the uncertainty often associated with sensitive catalytic processes.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the use of benign reagents facilitate easy scale-up from laboratory gram quantities to multi-ton commercial production volumes. The process generates minimal hazardous waste compared to traditional methods involving toxic metals, aligning with increasingly strict environmental regulations and corporate sustainability goals. This eco-friendly profile not only reduces the regulatory burden but also enhances the marketability of the final products to environmentally conscious clients in the global pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the process utilizes elemental iodine as a non-metallic promoter, completely avoiding the use of toxic heavy metals like palladium or copper, which simplifies purification and reduces regulatory burdens for API production.
Q: What are the advantages regarding raw material availability?
A: The method relies on commercially available aryl ethyl ketones and trifluoroethylimide hydrazides, which are cheap and easy to obtain, ensuring a stable and cost-effective supply chain for large-scale manufacturing.
Q: Is this process suitable for industrial scale-up?
A: Yes, the reaction conditions are mild, do not require strict anhydrous or oxygen-free environments, and have been demonstrated to scale easily from gram levels, making it highly viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, partnering with an experienced CDMO is crucial for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole delivered meets the highest industry standards for impurity control and chemical identity.
We invite you to collaborate with our technical procurement team to explore how this metal-free synthesis can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this iodine-promoted route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a competitive edge in the global market.
