Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand robust synthetic routes for fluorinated heterocycles, particularly 1,2,3-triazoles, due to their exceptional metabolic stability and bioavailability. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that addresses critical safety and efficiency bottlenecks found in legacy technologies. This innovation utilizes a base-promoted cyclization strategy involving trifluoroethylimidoyl chloride and diazo compounds, completely bypassing the need for transition metal catalysts or explosive azide reagents. For R&D directors and procurement specialists, this represents a paradigm shift towards safer, more cost-effective manufacturing of high-value intermediates used in beta-3 adrenergic receptor agonists and various functional materials.
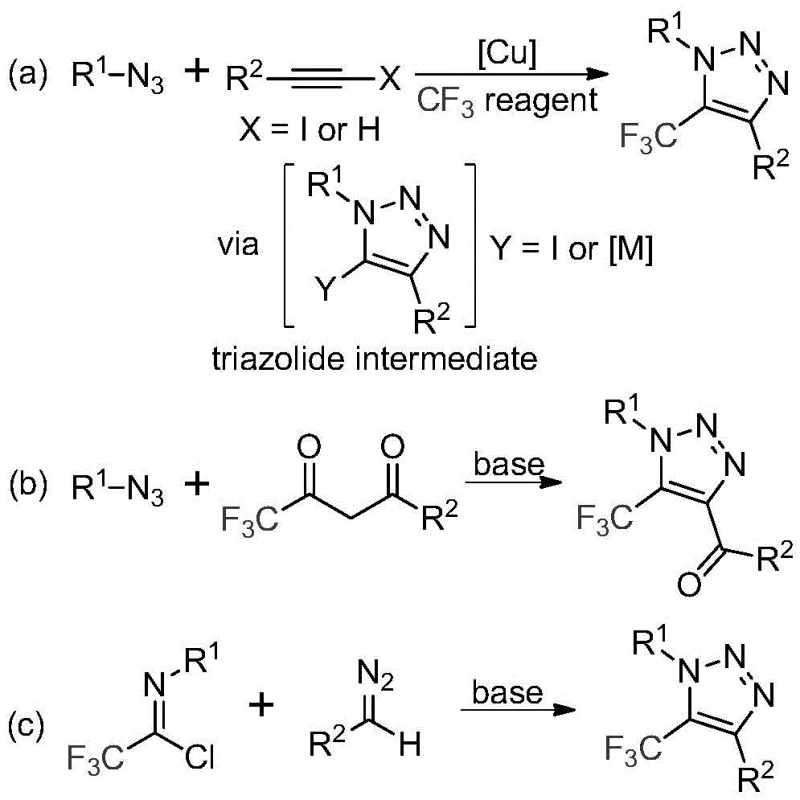
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed [3+2] cycloadditions between alkynes and organic azides, followed by trifluoromethylation steps. These traditional pathways suffer from severe drawbacks, primarily the reliance on toxic and potentially explosive organic azides, which pose significant safety hazards during scale-up and storage. Furthermore, the requirement for copper catalysts introduces complex downstream processing challenges, necessitating rigorous heavy metal removal steps to meet stringent pharmaceutical purity specifications. Alternative organocatalytic routes involving azides and trifluoromethyl ketones also inherit these safety liabilities while often struggling with limited substrate scope and moderate reaction efficiencies.
The Novel Approach
In stark contrast, the methodology disclosed in CN113121462B employs a metal-free, base-promoted strategy that utilizes cheap and readily available diazo compounds and trifluoroethylimidoyl chlorides as starting materials. This approach fundamentally alters the risk profile of the synthesis by eliminating explosive azides and toxic transition metals entirely. The reaction proceeds smoothly in common aprotic solvents like acetonitrile at moderate temperatures, offering a streamlined workflow that is inherently safer and more environmentally benign. By shifting the synthetic logic to a nucleophilic addition-elimination sequence followed by cyclization, this method provides a versatile platform for generating diverse 1,4-disubstituted triazoles with high functional group tolerance.
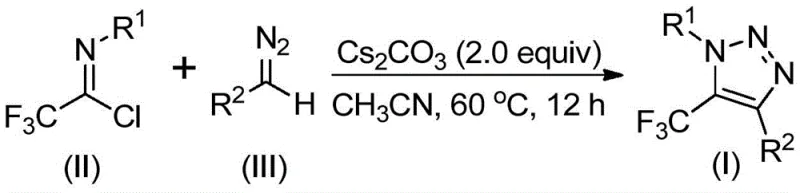
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation is distinct from classical click chemistry, relying on the unique reactivity of diazo compounds under basic conditions. The reaction is initiated by the deprotonation or activation of the diazo species by cesium carbonate, facilitating a nucleophilic attack on the electrophilic carbon of the trifluoroethylimidoyl chloride. This intermolecular addition-elimination process promotes the critical carbon-carbon bond formation necessary for the heterocyclic framework. Subsequently, the intermediate undergoes an intramolecular 5-endo-dig cyclization, driven by the electronic properties of the trifluoromethyl group and the nitrogen lone pairs, to close the triazole ring efficiently. This mechanism avoids the formation of stable metal-carbene intermediates, thereby simplifying the kinetic profile of the reaction.
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-complexed byproducts that are notoriously difficult to separate. The primary side reactions typically involve the decomposition of the diazo compound or hydrolysis of the imidoyl chloride, both of which can be effectively managed by controlling moisture levels and maintaining the optimal stoichiometric ratio of 1:1.5:2 (imidoyl chloride:diazo:base). The use of 4 Å molecular sieves further ensures anhydrous conditions, suppressing hydrolytic degradation and driving the equilibrium towards the desired triazole product. This clean reaction profile results in crude products that are easier to purify, directly impacting the overall yield and operational cost.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The experimental protocol outlined in the patent demonstrates a highly reproducible procedure suitable for both laboratory optimization and pilot plant operations. The process involves charging a reactor with cesium carbonate, the specific trifluoroethylimidoyl chloride, and the diazo compound in acetonitrile, followed by heating to 60°C for approximately 12 hours. Detailed standard operating procedures regarding mixing rates, addition sequences, and workup protocols are essential for maximizing yield and safety. For a comprehensive step-by-step guide tailored to your specific substrate, please refer to the technical documentation below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in acetonitrile solvent.
- Heat the reaction mixture to 60°C and stir for 12 hours under inert atmosphere.
- Filter the mixture, concentrate, and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers substantial strategic advantages beyond mere chemical novelty. The elimination of copper catalysts removes the need for expensive scavenging resins and extensive filtration processes, leading to significant reductions in raw material consumption and waste disposal costs. Furthermore, the avoidance of hazardous azide reagents simplifies regulatory compliance and lowers insurance premiums associated with handling explosive materials, creating a more resilient and cost-efficient supply chain.
- Cost Reduction in Manufacturing: The process utilizes cesium carbonate, a relatively inexpensive inorganic base, instead of costly transition metal catalysts or specialized ligands. By removing the heavy metal removal step, manufacturers can drastically reduce the number of unit operations required, lowering energy consumption and labor costs. The high atom economy of the cyclization step ensures that raw materials are converted efficiently into the final product, minimizing waste generation and maximizing return on investment for every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: The starting materials, specifically trifluoroethylimidoyl chlorides and diazo compounds, are derived from commodity chemicals and are widely available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks that often plague specialized catalyst-dependent syntheses. Additionally, the mild reaction conditions (60°C) allow for the use of standard glass-lined or stainless steel reactors without the need for exotic high-pressure or cryogenic equipment, ensuring that production can be easily scaled across different manufacturing sites globally.
- Scalability and Environmental Compliance: The metal-free nature of this reaction aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations regarding heavy metal discharge. Scaling this process from grams to tons does not introduce new safety hazards related to thermal runaways associated with azide accumulation, as the diazo compounds are consumed steadily in the presence of the base. This inherent safety margin facilitates faster technology transfer and regulatory approval, allowing companies to bring fluorinated drug candidates to market more rapidly while maintaining a sustainable environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method in a commercial setting. These answers are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a reliable foundation for process development decisions. Understanding these nuances is critical for R&D teams planning to integrate this technology into their existing manufacturing pipelines.
Q: Does this synthesis require toxic azide reagents?
A: No, unlike traditional methods, this patent describes a route using stable diazo compounds and imidoyl chlorides, eliminating the need for hazardous organic azides.
Q: What is the optimal temperature for this reaction?
A: The patent data indicates an optimal reaction temperature of 60°C in acetonitrile, balancing reaction rate and product stability.
Q: Is transition metal removal required?
A: No, the process is metal-free, utilizing cesium carbonate as a promoter, which significantly simplifies downstream purification and reduces heavy metal contamination risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole delivered meets the highest industry standards for potency and safety.
We invite you to collaborate with our technical team to explore how this innovative process can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project's unique needs, ensuring a competitive edge in the global market.
