Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial API Production
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial API Production
The pharmaceutical and agrochemical industries are constantly seeking robust, safe, and cost-effective pathways to construct nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous traditional methodologies, offering a streamlined, metal-free, and azide-free synthetic route that utilizes cheap and readily available starting materials. For R&D directors and procurement managers alike, this innovation addresses critical pain points regarding safety, impurity profiles, and raw material costs, positioning it as a highly attractive candidate for the reliable pharmaceutical intermediate supplier market. The method employs a base-promoted cyclization strategy that not only simplifies the operational workflow but also ensures high reaction efficiency, making it an ideal solution for cost reduction in API manufacturing where margin compression is a constant challenge.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which carry substantial baggage for large-scale production. The first common approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation; this pathway is fraught with safety risks due to the inherent instability and explosivity of organic azides, alongside the regulatory and environmental burden of removing toxic copper residues from the final active pharmaceutical ingredient. The second conventional route utilizes organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones, which similarly suffers from the safety liabilities associated with handling bulk quantities of azide reagents. Furthermore, these traditional methods often require specialized trifluoromethylating reagents that are expensive and difficult to source consistently, creating bottlenecks for the supply chain head who must guarantee continuity of supply. The reliance on transition metals also introduces complex purification steps, such as scavenging and extensive chromatography, which drastically increase the overall cost of goods sold and extend lead times for high-purity intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in patent CN113121462B introduces a remarkably simple yet powerful alternative that circumvents the use of both toxic azides and expensive transition metal catalysts. This novel approach leverages a base-promoted reaction between readily accessible trifluoroethylimidoyl chlorides and diazo compounds to directly forge the triazole ring with high regioselectivity. By eliminating the need for copper catalysts, the process inherently produces a cleaner crude reaction profile, significantly reducing the burden on downstream purification teams and lowering the risk of metal contamination in the final drug substance. The use of cesium carbonate as a mild promoter allows the reaction to proceed under relatively gentle thermal conditions, typically between 50°C and 70°C, which enhances energy efficiency and operational safety compared to high-temperature alternatives. This strategic pivot not only mitigates the safety risks associated with explosive azides but also opens up a broader substrate scope, allowing for the facile introduction of diverse functional groups at the N1 and C4 positions of the triazole core.
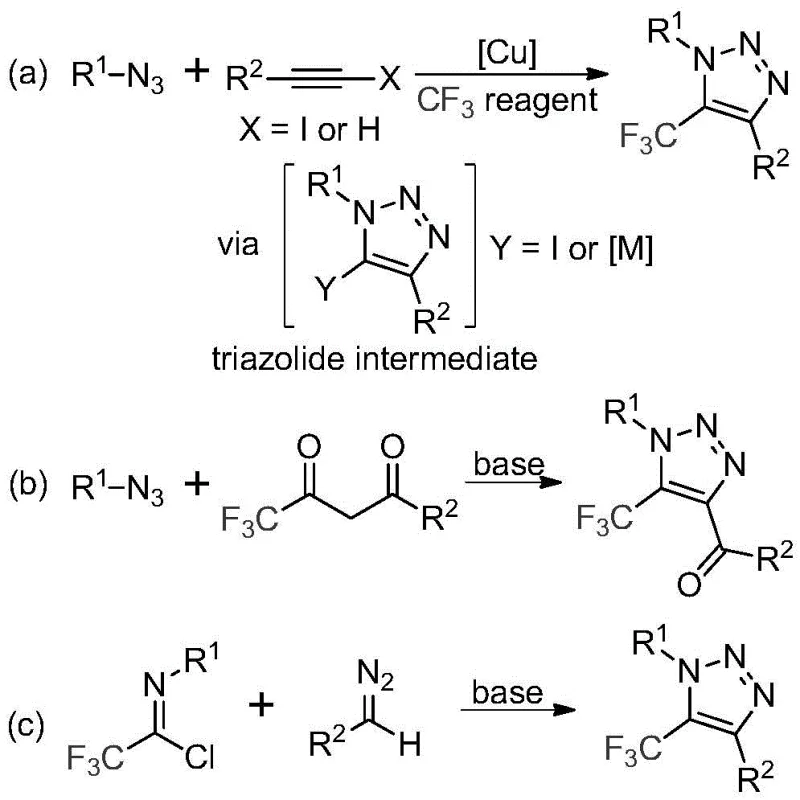
The core transformation is elegantly depicted in the reaction scheme below, highlighting the direct convergence of the imidoyl chloride and diazo precursors to form the target scaffold without intermediate metal complexes.
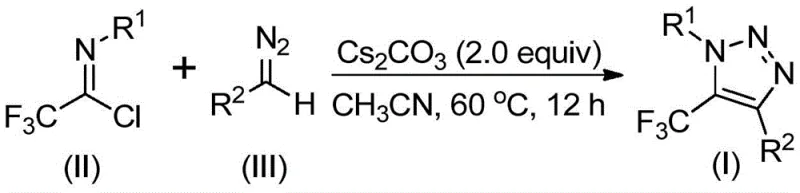
Mechanistic Insights into Base-Promoted Cyclization
From a mechanistic perspective, this transformation offers a fascinating glimpse into how simple base promotion can drive complex heterocycle formation without the need for redox-active metals. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process, where the diazo compound acts as a nucleophile attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial step facilitates the critical carbon-carbon bond formation that sets the stage for ring closure, generating a reactive intermediate that is primed for cyclization. Subsequently, the system undergoes an intramolecular 5-endo-dig cyclization, a process that is thermodynamically driven by the formation of the aromatic triazole ring and the expulsion of the chloride leaving group. This mechanism is distinct from the stepwise metallacycle formation seen in copper-catalyzed variants, thereby avoiding the potential for side reactions associated with metal coordination and beta-hydride elimination.
Understanding this mechanism is crucial for R&D teams focused on impurity control, as the absence of metal catalysts eliminates a major source of trace impurities that often plague API batches. The clean nature of the base-promoted pathway means that the primary impurities are likely to be unreacted starting materials or simple hydrolysis products, which are generally easier to separate via standard crystallization or chromatography techniques than metal-chelated byproducts. Furthermore, the tolerance of the reaction conditions towards various substituents on the aryl rings of both the imidoyl chloride and the diazo compound suggests a robust electronic flexibility. This allows chemists to fine-tune the electronic properties of the final molecule—such as introducing electron-withdrawing fluorine atoms or electron-donating methoxy groups—without compromising the yield or necessitating a complete re-optimization of the catalytic system, ensuring consistent quality across different analogues.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and solvent choices to maximize yield and purity. The patent data indicates that while various aprotic solvents can support the reaction, acetonitrile stands out as the optimal medium, providing superior solubility for the reagents and promoting higher conversion rates compared to toluene or dioxane. The molar ratio of reactants is also a critical parameter, with a slight excess of the diazo compound relative to the imidoyl chloride recommended to drive the reaction to completion, given the potentially labile nature of the diazo species. Detailed standard operating procedures regarding temperature ramping, addition rates, and workup protocols are essential for reproducibility, and the full standardized synthesis steps are outlined in the guide below for technical reference.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages that go beyond mere chemical elegance. The shift away from specialized, hazardous reagents like organic azides and expensive copper catalysts fundamentally alters the cost structure and risk profile of the manufacturing process. By utilizing commodity chemicals such as cesium carbonate and widely available imidoyl chlorides, the dependency on niche suppliers is reduced, thereby enhancing supply chain resilience against market volatility. Moreover, the simplification of the workup procedure—often requiring only filtration and standard chromatography rather than complex metal scavenging—drastically reduces the consumption of auxiliary materials and processing time, leading to substantial cost savings in the overall production budget.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive ligand systems and the subsequent rigorous purification steps required to meet strict residual metal limits in pharmaceuticals. This qualitative improvement in process efficiency directly correlates to a lower cost of goods, as the facility can achieve higher throughput with fewer unit operations and reduced waste disposal costs associated with heavy metal contaminants. Additionally, the use of cesium carbonate, a relatively inexpensive inorganic base, further drives down raw material expenses compared to proprietary organocatalysts or rare-earth metal complexes often used in alternative trifluoromethylation strategies.
- Enhanced Supply Chain Reliability: Sourcing stable, non-explosive starting materials significantly de-risks the procurement process, as diazo compounds and imidoyl chlorides are generally more stable and easier to transport than sensitive azide reagents. This stability ensures that inventory can be held safely for longer periods without degradation, allowing for better demand planning and reducing the likelihood of production stoppages due to reagent spoilage or shipping restrictions on hazardous materials. The broad availability of these precursors from multiple global chemical suppliers also prevents single-source bottlenecks, ensuring a continuous flow of materials for commercial scale-up of complex heterocycles.
- Scalability and Environmental Compliance: The mild reaction conditions and metal-free nature of this process align perfectly with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. Scaling this reaction from gram to kilogram levels does not introduce the exponential safety risks associated with scaling azide chemistry, making it a safer choice for contract manufacturing organizations (CMOs) looking to expand capacity. The reduced generation of heavy metal waste streams simplifies effluent treatment processes, lowering the environmental footprint and associated compliance costs for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, derived directly from the experimental data and beneficial effects described in the intellectual property documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this metal-free methodology for your specific project needs. Understanding these nuances is vital for making informed decisions about process development and vendor selection.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on toxic and explosive organic azides or heavy metal catalysts like copper. This patented process utilizes stable diazo compounds and imidoyl chlorides under metal-free conditions, drastically reducing safety hazards and eliminating the need for expensive heavy metal scavenging steps.
Q: What are the typical reaction conditions for this transformation?
A: The reaction operates under mild thermal conditions, typically between 50°C and 70°C, using cesium carbonate as a promoter in acetonitrile. This avoids the extreme temperatures or pressures often required in older cycloaddition protocols.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent explicitly demonstrates that the method can be easily extended to gram-scale synthesis with high efficiency. The use of commercially available starting materials and simple workup procedures makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for accelerating the development of next-generation therapeutics and agrochemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate delivered meets the highest global standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can be tailored to your specific pipeline requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in delivering high-value chemical solutions.
