Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
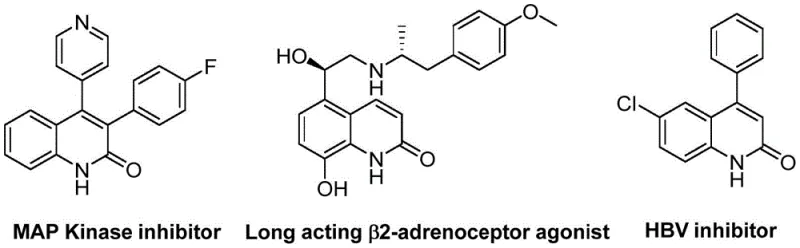
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing privileged heterocyclic scaffolds. Patent CN113045489B introduces a groundbreaking preparation method for 3-arylquinolin-2(1H) ketone derivatives, a class of compounds renowned for their presence in diverse bioactive molecules and natural products. As illustrated in the structural diversity of biologically active targets below, these quinolinone cores are foundational to MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors, underscoring their critical value in modern drug discovery pipelines. This novel synthetic route leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm from traditional, hazardous methods to a safer, more efficient protocol.
The significance of this technology lies in its ability to utilize benzisoxazole as a dual-purpose reagent, acting simultaneously as the nitrogen source and the formyl source. This innovation addresses long-standing challenges in heterocycle synthesis, offering a streamlined pathway that is not only operationally simple but also demonstrates exceptional substrate compatibility. For R&D directors and procurement managers alike, this represents a tangible opportunity for cost reduction in pharmaceutical intermediate manufacturing, as it replaces expensive or dangerous reagents with readily available, stable solids. The method's high efficiency and broad applicability make it an ideal candidate for integration into existing supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods have served the industry for decades, they are increasingly viewed as suboptimal for modern commercial scale-up due to significant drawbacks. Conventional approaches often require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to poor functional group tolerance and the formation of complex impurity profiles that are difficult to remove. Furthermore, many traditional carbonylation strategies necessitate the use of toxic carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure equipment that increases capital expenditure. These limitations result in higher operational costs, extended lead times for high-purity intermediates, and environmental compliance burdens that strain supply chain reliability.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113045489B offers a sophisticated solution through a palladium-catalyzed aminocarbonylation reaction. As depicted in the general reaction scheme below, this approach couples benzisoxazole derivatives with benzyl chloride compounds under mild conditions using a Pd(OAc)2/(S)-BINAP catalyst system. The use of molybdenum hexacarbonyl [Mo(CO)6] as a solid CO surrogate eliminates the need for handling gaseous carbon monoxide, drastically improving workplace safety and simplifying reactor requirements. This novel route operates at a moderate temperature of 100°C in DME solvent, utilizing triethylamine as a base and water as a crucial additive to facilitate the transformation. The result is a highly efficient process that delivers target quinolinones with excellent yields, often exceeding 90%, while maintaining a clean reaction profile that simplifies downstream purification.
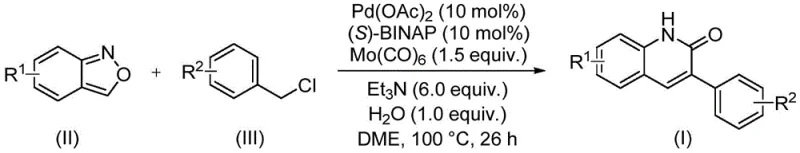
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The success of this synthetic strategy hinges on the intricate interplay between the palladium catalyst and the unique reactivity of the benzisoxazole ring. Mechanistically, the reaction initiates with the oxidative addition of the benzyl chloride to the active Pd(0) species generated in situ from Pd(OAc)2 and the chiral ligand (S)-BINAP. Simultaneously, the benzisoxazole ring undergoes activation, likely facilitated by the basic conditions and the presence of water, which assists in the ring-opening or N-O bond cleavage to release the necessary nitrogen and carbonyl fragments. The molybdenum hexacarbonyl serves as a controlled source of carbon monoxide, which inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This step is critical for constructing the lactam carbonyl of the quinolinone core. Subsequent intramolecular cyclization and reductive elimination release the final 3-arylquinolin-2(1H)-one product and regenerate the catalytic species, completing the cycle.
From an impurity control perspective, this mechanism offers distinct advantages over random condensation reactions. The specificity of the palladium catalyst ensures that the carbonylation occurs selectively at the benzylic position, minimizing side reactions such as homocoupling of the benzyl chloride or polymerization of the intermediates. The broad functional group tolerance observed in the patent examples, ranging from electron-donating methoxy groups to electron-withdrawing cyano and trifluoromethyl substituents, confirms the robustness of the catalytic cycle. As shown in the specific examples below, the method consistently produces well-defined structures like compounds (I-1) through (I-5) with high purity. This predictability is vital for regulatory compliance in API manufacturing, where consistent impurity profiles are mandatory for batch-to-batch reproducibility and patient safety.
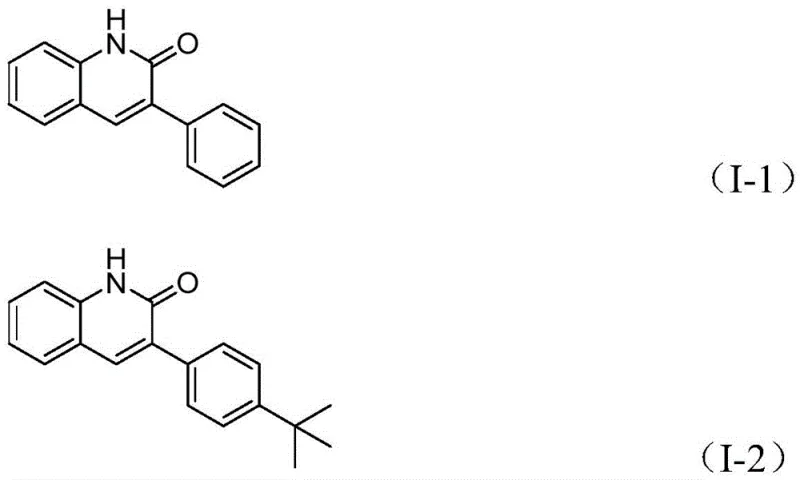
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction parameters to maximize yield and purity. The standard protocol involves charging a sealed reaction vessel with the palladium catalyst, ligand, carbonyl source, base, and substrates in a specific molar ratio, typically favoring an excess of the benzyl chloride to drive the reaction to completion. The mixture is then heated to reflux in DME for a defined period, usually around 26 hours, to ensure full conversion of the starting materials. Following the reaction, a straightforward workup procedure involving filtration and silica gel treatment allows for the isolation of the crude product, which can be further purified by column chromatography if necessary. For detailed operational parameters and specific stoichiometric guidelines, please refer to the standardized synthesis steps outlined below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C in a sealed tube and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into immediate strategic benefits regarding cost, safety, and scalability. By replacing hazardous gaseous reagents with stable solid alternatives like Mo(CO)6 and benzisoxazole, the process significantly reduces the infrastructure costs associated with high-pressure gas handling and safety monitoring systems. This shift not only lowers the barrier to entry for manufacturing but also enhances supply chain reliability by utilizing raw materials that are commodity chemicals with stable global availability. The simplified workup and high reaction efficiency mean that production cycles can be shortened, reducing the overall lead time for high-purity pharmaceutical intermediates and allowing for more responsive inventory management in a volatile market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and dangerous carbon monoxide gas cylinders, which require specialized storage and delivery logistics. Instead, the use of solid molybdenum hexacarbonyl and inexpensive benzisoxazole derivatives drastically simplifies the bill of materials. Furthermore, the high yields reported in the patent data, often reaching up to 97%, imply a substantial reduction in raw material waste and solvent consumption per kilogram of product. This efficiency directly correlates to a lower cost of goods sold (COGS), providing a competitive margin advantage for manufacturers producing complex heterocyclic intermediates at scale.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials such as benzyl chlorides and substituted benzisoxazoles, which are produced by numerous chemical suppliers globally. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague specialized reagent markets. Additionally, the robustness of the reaction conditions—tolerating moisture and air to a reasonable extent compared to sensitive organometallic protocols—reduces the likelihood of batch failures due to minor environmental fluctuations. This reliability ensures consistent delivery schedules, a critical factor for downstream API manufacturers who depend on just-in-time inventory flows.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by the absence of high-pressure gas feeds, allowing the use of standard glass-lined or stainless steel reactors without the need for autoclaves rated for toxic gases. The environmental footprint is also significantly reduced; avoiding CO gas minimizes the risk of accidental releases, and the high atom economy of the transformation reduces the volume of chemical waste generated. This alignment with green chemistry principles simplifies environmental permitting and waste disposal processes, ensuring long-term operational continuity in regions with stringent ecological regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your current manufacturing portfolio.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate carbonylation reagents like toxic carbon monoxide gas and simplifying the reaction stoichiometry.
Q: What is the functional group tolerance of this Pd-catalyzed method?
A: The method exhibits excellent functional group tolerance, successfully accommodating substituents such as methoxy, chloro, cyano, trifluoromethyl, and tert-butyl groups on both the benzisoxazole and benzyl chloride substrates.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available starting materials and avoids hazardous gaseous reagents, making it highly suitable for safe and scalable commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN113045489B for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data for related quinolinone derivatives and to discuss comprehensive route feasibility assessments that will secure your supply chain for the future.
