Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic scaffolds that serve as the backbone for potent bioactive molecules. Patent CN113045489A introduces a groundbreaking preparation method for 3-arylquinoline-2(1H)-ketone derivatives, a class of compounds renowned for their diverse biological activities ranging from MAP Kinase inhibition to antiviral properties. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by employing a palladium-catalyzed aminocarbonylation strategy that utilizes benzisoxazole as a unique dual nitrogen and formyl source. The significance of this technology lies in its ability to streamline the construction of the quinolinone core, which is a privileged structure found in numerous therapeutic agents such as long-acting β2-adrenoceptor agonists and HBV inhibitors. By leveraging this novel pathway, manufacturers can access complex molecular architectures with enhanced operational simplicity and superior atom economy compared to legacy methods.
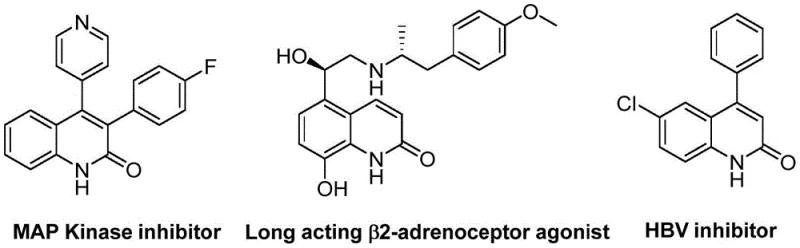
The strategic value of this patent extends beyond mere academic interest; it offers a tangible solution for reliable pharmaceutical intermediate supplier networks aiming to secure stable supplies of high-value building blocks. The method described allows for the synthesis of a broad spectrum of derivatives by simply varying the substituents on the benzisoxazole and benzyl chloride starting materials. This modularity is crucial for medicinal chemistry campaigns where rapid analog generation is required to optimize potency and selectivity. Furthermore, the reaction conditions are remarkably mild and tolerant of various functional groups, ensuring that sensitive moieties required for downstream biological activity remain intact throughout the synthesis. This level of functional group compatibility is a hallmark of modern process chemistry and positions this technology as a cornerstone for next-generation drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)-one derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods have served the industry for decades, they are often plagued by significant drawbacks that hinder efficient large-scale production. Traditional routes frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Moreover, many conventional processes involve multi-step sequences that necessitate the isolation of unstable intermediates, thereby increasing overall processing time and reducing cumulative yield. The reliance on hazardous reagents, such as gaseous carbon monoxide in carbonylation approaches, also introduces severe safety concerns and requires specialized equipment for containment and handling, driving up capital expenditure and operational complexity for manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated palladium-catalyzed system that fundamentally reimagines the construction of the quinolinone ring. By employing benzisoxazole as both the nitrogen source and the formyl source, the method elegantly bypasses the need for external carbon monoxide gas, significantly enhancing process safety and operational ease. The reaction proceeds through a concerted mechanism where the benzisoxazole ring opens under catalytic conditions to provide the necessary atoms for ring closure, coupled with a benzyl chloride compound to introduce the aryl substituent at the 3-position. This one-pot transformation not only reduces the number of unit operations but also minimizes waste generation, aligning perfectly with green chemistry principles. The use of readily available starting materials like benzisoxazole and substituted benzyl chlorides ensures that the supply chain remains robust and cost-effective, making this method highly attractive for commercial adoption in the fine chemical sector.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The core of this technological breakthrough lies in the intricate catalytic cycle driven by the palladium complex. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step is critical as it activates the carbon-chlorine bond, setting the stage for subsequent carbonyl insertion. Uniquely, the carbon monoxide required for the carbonylation step is not supplied externally but is released from molybdenum hexacarbonyl (Mo(CO)6) under the reaction conditions, acting as a safe and controllable CO surrogate. The coordination of the benzisoxazole to the metal center facilitates the ring-opening process, effectively delivering the nitrogen and carbonyl fragments needed to construct the lactam ring of the quinolinone. This mechanistic pathway ensures high regioselectivity and minimizes side reactions, resulting in the clean formation of the desired 3-arylquinolin-2(1H)-one scaffold.
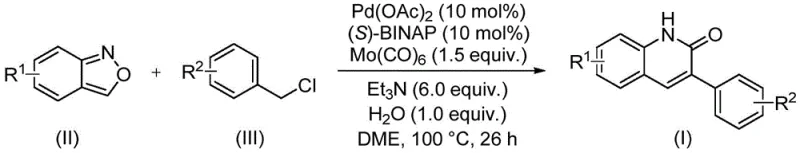
Understanding the impurity control mechanism is vital for R&D directors focused on product quality. The presence of triethylamine as a base and water as an additive plays a pivotal role in stabilizing the catalytic cycle and promoting the hydrolysis steps necessary for product release. The specific ratio of reagents, particularly the 0.1:0.1:6:1.0 molar ratio of palladium catalyst, ligand, triethylamine, and water, has been optimized to suppress the formation of homocoupling byproducts and unreacted starting materials. The reaction temperature of 100 °C provides sufficient energy to overcome activation barriers without inducing thermal decomposition of the sensitive heterocyclic products. Post-reaction processing involves simple filtration and silica gel treatment followed by column chromatography, which effectively removes trace metal residues and organic impurities, ensuring the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility in a GMP environment. The procedure involves charging a sealed reactor with the precise stoichiometric amounts of palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative in dimethoxyethane (DME) solvent. The mixture is then heated to 100 °C and maintained for 26 hours to ensure full conversion, as evidenced by the high yields reported across fifteen different examples in the patent data. This standardized protocol allows for the consistent production of various derivatives, including those with electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups like cyano and chloro. For detailed operational parameters and specific substrate variations, please refer to the structured guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum carbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100 °C and stir continuously for 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented method offers substantial strategic benefits that directly impact the bottom line and supply security. The shift towards using benzisoxazole and benzyl chlorides as primary feedstocks leverages a supply chain that is well-established and globally accessible, reducing the risk of raw material shortages that often plague specialty chemical manufacturing. By eliminating the need for high-pressure carbon monoxide cylinders and the associated safety infrastructure, facilities can significantly lower their capital investment and insurance costs, leading to a more favorable cost structure for the final intermediate. Furthermore, the high reaction efficiency and broad substrate tolerance mean that a single production line can be easily adapted to manufacture a wide library of quinolinone derivatives, maximizing asset utilization and flexibility in response to changing market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and commercially available catalysts and ligands, such as palladium acetate and (S)-BINAP, which are used in relatively low loading levels. The elimination of hazardous gas handling systems and the simplification of the workup procedure to a standard filtration and chromatography sequence drastically reduce operational expenditures. Additionally, the high yields observed, often exceeding 90% for many substrates, minimize raw material waste and maximize the output per batch, contributing to significant overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, solid, or liquid reagents rather than gaseous ones enhances the logistical simplicity of the supply chain. Benzisoxazole and substituted benzyl chlorides are commodity chemicals with multiple global suppliers, ensuring that procurement teams can negotiate competitive pricing and secure long-term contracts without fear of single-source dependency. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor fluctuations in environmental parameters or reagent quality, guaranteeing consistent delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in sealed tube experiments that mimic the conditions of larger autoclaves used in pilot and commercial plants. The use of DME as a solvent and the absence of heavy metal waste streams beyond the recoverable palladium catalyst align with increasingly strict environmental regulations. This compliance reduces the burden on waste treatment facilities and lowers the environmental footprint of the manufacturing process, making it a sustainable choice for companies committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational details. These insights are derived directly from the experimental data provided in the patent, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for integrating this methodology into existing production workflows and for assessing its fit within your current portfolio of synthetic capabilities.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves as a dual nitrogen and formyl source, eliminating the need for separate carbon monoxide gas handling and simplifying the reaction setup while maintaining high efficiency.
Q: What is the typical yield range for this palladium-catalyzed process?
A: According to patent data, the reaction demonstrates high efficiency with isolated yields ranging from 68% to 97% across various substituted substrates.
Q: Is this method suitable for large-scale pharmaceutical production?
A: Yes, the method utilizes commercially available raw materials and standard purification techniques like column chromatography, making it highly scalable for industrial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed synthesis for the pharmaceutical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe management of palladium catalysts and the rigorous purification steps necessary to achieve stringent purity specifications. With our dedicated rigorous QC labs, we guarantee that every batch of 3-arylquinolin-2(1H)-one derivative meets the highest standards of quality and consistency required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with confidence and reliability.
