Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical cores in drug discovery. Patent CN113045489B introduces a groundbreaking methodology for the preparation of 3-arylquinolin-2(1H)-one derivatives, a class of compounds renowned for their diverse biological activities ranging from antibiotic to antitumor properties. This innovation leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally alters the traditional approach to constructing the quinolinone ring system. By utilizing benzisoxazole as a dual-purpose synthon acting as both the nitrogen and formyl source, the process achieves remarkable atom economy and operational simplicity. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-value pharmaceutical intermediates. The technology not only addresses the limitations of classical cyclization methods but also offers a versatile platform capable of accommodating a wide array of functional groups, thereby facilitating the rapid synthesis of diverse analog libraries for structure-activity relationship (SAR) studies.
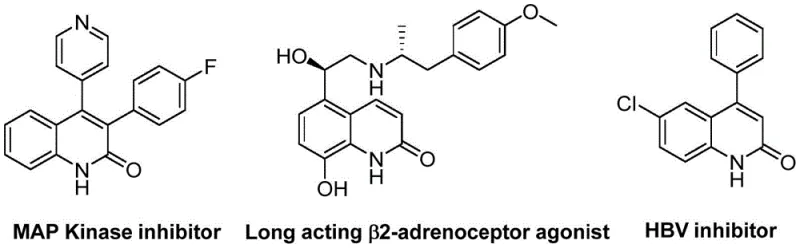
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)-one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods are well-documented in academic literature, they often suffer from significant drawbacks when applied to modern industrial settings. Traditional routes frequently require harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to the degradation of sensitive functional groups and complicate the isolation of pure products. Furthermore, these conventional processes often exhibit poor atom economy, generating substantial amounts of chemical waste that necessitate costly disposal protocols. The reliance on multi-step sequences to install the necessary nitrogen and carbonyl functionalities also increases the overall production time and capital expenditure. For a reliable pharmaceutical intermediate supplier, these inefficiencies translate into higher costs and longer lead times, creating bottlenecks in the development of new therapeutic agents.
The Novel Approach
In stark contrast, the novel palladium-catalyzed aminocarbonylation method described in the patent data offers a streamlined and efficient alternative. This approach utilizes readily available benzisoxazole and benzyl chloride compounds as starting materials, reacting them under mild conditions to directly forge the quinolinone core. The reaction proceeds at a moderate temperature of 100°C in ethylene glycol dimethyl ether (DME), utilizing a catalytic system composed of palladium acetate, (S)-BINAP, and molybdenum hexacarbonyl. This transition metal-catalyzed pathway demonstrates exceptional functional group tolerance, allowing for the incorporation of diverse substituents such as halogens, alkoxy groups, and cyano groups without the need for extensive protecting group strategies. The ability to synthesize a variety of 3-arylquinolin-2(1H)-one derivatives in a single pot significantly enhances the practicality of the method, making it an ideal candidate for cost reduction in API manufacturing where speed and flexibility are paramount.
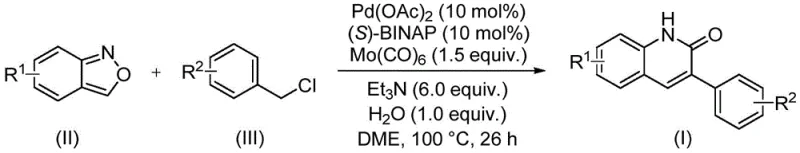
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The mechanistic elegance of this transformation lies in the unique reactivity of the benzisoxazole ring under palladium catalysis. Unlike traditional amide couplings that require pre-functionalized amines and acid chlorides, this system exploits the inherent strain and electronic properties of the benzisoxazole moiety. The palladium catalyst, ligated by the chiral (S)-BINAP phosphine, facilitates the oxidative addition into the benzyl chloride, forming a reactive organopalladium species. Simultaneously, the benzisoxazole undergoes ring opening or activation, serving as an internal source of nitrogen and the formyl group required for the lactam formation. The presence of molybdenum hexacarbonyl likely assists in the carbonylation step, providing the necessary carbon monoxide equivalents or stabilizing the metal center during the catalytic cycle. This intricate interplay between the transition metal catalyst and the heterocyclic substrate ensures high regioselectivity and minimizes the formation of undesired side products, which is critical for maintaining high purity standards in high-purity pharmaceutical intermediates.
Furthermore, the reaction conditions are meticulously optimized to balance reactivity and stability. The use of triethylamine as a base and a stoichiometric amount of water plays a crucial role in proton management and hydrolysis steps within the catalytic cycle. The tolerance for various substituents on both the benzisoxazole (R1) and the benzyl chloride (R2) rings indicates a robust catalytic cycle that is not easily poisoned by electron-withdrawing or electron-donating groups. For process chemists, understanding this mechanism is vital for troubleshooting and optimizing the reaction for commercial scale-up of complex pharmaceutical intermediates. The high yields reported, often exceeding 90% for unsubstituted or simply substituted substrates, underscore the efficiency of this catalytic system in converting inexpensive starting materials into valuable heterocyclic building blocks with minimal waste generation.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating these valuable derivatives. The procedure involves charging a sealed reaction vessel with the palladium catalyst, ligand, carbonyl source, base, water, and the two primary organic substrates. The mixture is then heated to facilitate the coupling reaction, followed by a straightforward workup procedure involving filtration and chromatography. This simplicity is a key advantage for manufacturing teams looking to implement new processes quickly.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride derivative.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture to ensure homogeneity before heating.
- Heat the reaction mixture to 100°C for 26 hours, then filter, mix with silica gel, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit stems from the utilization of cheap and readily available starting materials. Benzisoxazole and substituted benzyl chlorides are commodity chemicals that can be sourced from multiple suppliers globally, ensuring supply chain resilience and reducing the risk of raw material shortages. The elimination of exotic or highly specialized reagents further simplifies the procurement process, allowing for better negotiation leverage and substantial cost savings in raw material acquisition. Additionally, the operational simplicity of the reaction, which requires only standard heating and stirring equipment, lowers the barrier to entry for contract manufacturing organizations (CMOs) and internal production facilities alike.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by consolidating multiple synthetic steps into a single catalytic transformation. By using benzisoxazole as a dual nitrogen and formyl source, the need for purchasing and handling separate amine and carbonyl reagents is eliminated, which reduces both material costs and inventory complexity. Furthermore, the high reaction efficiency and yield minimize the loss of valuable intermediates, leading to a more economical use of resources. The mild reaction conditions also contribute to lower energy consumption compared to high-temperature or high-pressure alternatives, resulting in reduced utility costs over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against various functional groups means that the same core process can be applied to synthesize a wide range of derivatives without needing to requalify entirely new synthetic routes. This flexibility allows supply chain managers to respond rapidly to changes in demand for specific analogs. Since the reagents are stable and commercially available, the risk of supply disruption is minimized. The straightforward post-processing, which involves standard filtration and chromatography, ensures that the turnaround time from reaction completion to final product release is kept to a minimum, enhancing overall supply chain agility.
- Scalability and Environmental Compliance: The reaction operates in ethylene glycol dimethyl ether (DME), a solvent that is manageable on a large scale, and the catalyst loading is kept relatively low, which reduces the burden of heavy metal waste disposal. The high atom economy of the reaction means less chemical waste is generated per kilogram of product, aligning with modern green chemistry principles and environmental regulations. This makes the process highly attractive for reducing lead time for high-purity pharmaceutical intermediates while maintaining compliance with increasingly stringent environmental standards, ensuring long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for potential adoption.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate amine and carbonyl reagents. This simplifies the reaction stoichiometry, reduces raw material costs, and streamlines the purification process by minimizing byproduct formation.
Q: Is this catalytic system suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available catalysts like palladium acetate and operates at a moderate temperature of 100°C. The robustness of the catalytic cycle and the wide functional group tolerance make it highly adaptable for scaling from laboratory grams to multi-ton commercial production.
Q: What is the typical purity profile of the resulting quinolinone derivatives?
A: The method demonstrates high reaction efficiency with yields often exceeding 90% for various substrates. Post-reaction processing involves standard filtration and column chromatography, which effectively removes catalyst residues and unreacted starting materials, ensuring high-purity intermediates suitable for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed aminocarbonylation described in CN113045489B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale optimization to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-arylquinolin-2(1H)-one derivative meets the highest quality standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can accelerate your path to market while optimizing your overall production costs.
