Scalable Synthesis of 2-Trifluoromethyl Imidazoles via Mild Palladium-Catalyzed Carbonylation for Commercial API Production
Introduction to Advanced Imidazole Synthesis Technology
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes for complex heterocycles. A significant breakthrough in this domain is documented in Chinese Patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. These structural motifs are ubiquitous in modern medicinal chemistry, serving as critical scaffolds in numerous bioactive molecules, including potential antiviral agents, kinase inhibitors, and histamine receptor antagonists. The introduction of a trifluoromethyl group into the imidazole core significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these compounds highly desirable for drug discovery programs. As a leading entity in fine chemical synthesis, understanding and leveraging such patented methodologies allows us to offer superior reliable pharmaceutical intermediate supplier capabilities to our global partners.
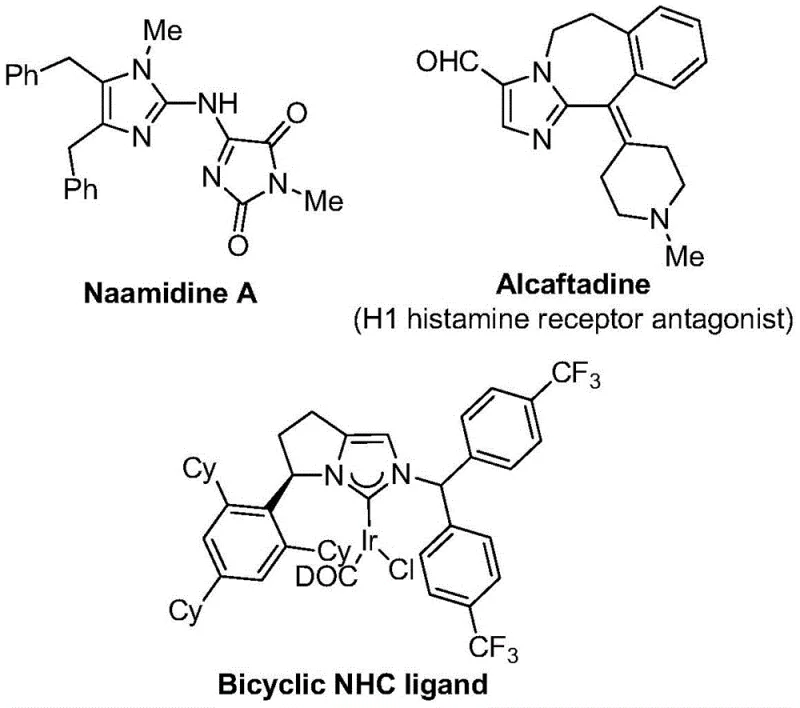
The strategic value of this technology lies not just in the final product quality but in the robustness of the process itself. Traditional methods for synthesizing nitrogen-containing heterocycles with trifluoromethyl functional groups often rely on specialized synthons like trifluorodiazoethane, which can be hazardous and difficult to handle on a large scale. In contrast, the methodology described in CN111423381B utilizes a transition metal palladium-catalyzed carbonylation series reaction. This approach transforms simple, commercially available starting materials—specifically trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts—into high-value 2-trifluoromethyl imidazoles. By shifting the paradigm towards milder conditions and safer reagents, this innovation addresses critical pain points for cost reduction in API manufacturing, enabling the production of high-purity pharmaceutical intermediates with improved safety profiles and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted imidazole rings has been fraught with synthetic challenges that hinder efficient commercialization. Conventional routes frequently necessitate the use of unstable or explosive reagents, such as diazo compounds, which pose significant safety risks during scale-up operations. Furthermore, many existing protocols require harsh reaction conditions, including elevated temperatures and strong bases, which can lead to poor functional group tolerance and the formation of complex impurity profiles. These limitations often result in low overall yields and necessitate extensive purification steps, driving up the cost of goods sold (COGS) and extending lead times. For procurement managers and supply chain heads, these inefficiencies translate into volatile pricing and unreliable delivery schedules for critical building blocks. Additionally, the reliance on high-pressure carbon monoxide gas in traditional carbonylation reactions introduces severe engineering controls and safety infrastructure requirements, further complicating the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
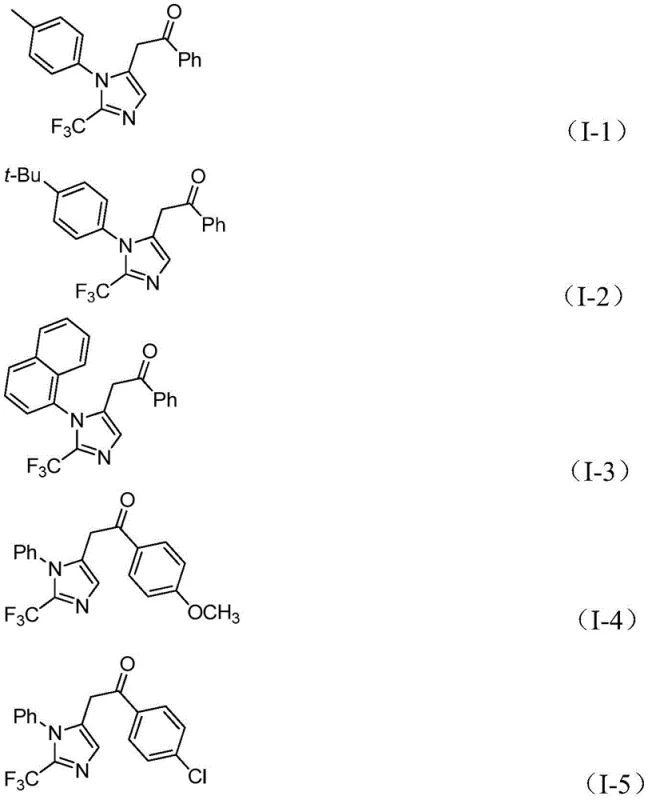
The methodology presented in patent CN111423381B offers a transformative solution by employing a palladium-catalyzed multicomponent reaction that operates under remarkably mild conditions. The core of this innovation is the use of formic acid and acetic anhydride as an in situ source of carbon monoxide, effectively eliminating the need for handling toxic CO gas cylinders. The reaction proceeds efficiently at a temperature of merely 30°C, utilizing tetrahydrofuran (THF) as a solvent and sodium bicarbonate as a mild base. This gentle environment ensures excellent compatibility with a wide range of functional groups, including halogens, nitro groups, and alkyl substituents, as demonstrated by the diverse substrate scope in the patent examples. By integrating trifluoroethylimidoyl chloride and propargylamine with diaryl iodonium salts, the process achieves high conversion rates and yields ranging from 65% to 97% across various substrates. This robustness makes it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates, as the simplified workup and purification procedures significantly accelerate the path from bench to plant.
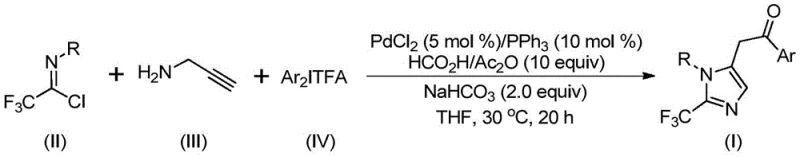
Mechanistic Insights into Palladium-Catalyzed Carbonylation
For R&D directors focused on process feasibility and impurity control, understanding the mechanistic underpinnings of this transformation is crucial. The reaction initiates with the formation of a trifluoroacetamidine intermediate through an intermolecular carbon-nitrogen bond promotion facilitated by the alkaline conditions. This intermediate subsequently undergoes isomerization to align the reactive centers for cyclization. The palladium catalyst, generated from PdCl2 and triphenylphosphine, then engages in a palladation of the alkyne moiety of the propargylamine derivative, forming a key alkenyl palladium intermediate. This species undergoes a second isomerization to generate an alkyl palladium intermediate, positioning the metal center for the subsequent carbonylation step. The carbon monoxide, released slowly and safely from the formic acid/acetic anhydride mixture, inserts into the palladium-carbon bond to create an acyl palladium intermediate. Finally, the diaryl iodonium salt participates via oxidative addition to form a tetravalent palladium species, which undergoes reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst. This intricate cycle ensures high atom economy and minimizes the formation of side products associated with radical pathways.
From an impurity control perspective, the use of diaryl iodonium salts as arylating agents is particularly advantageous. Unlike traditional cross-coupling partners that might leave behind difficult-to-remove organometallic residues, the byproducts of iodonium salt decomposition are typically iodobenzene derivatives, which are often easier to separate via standard chromatographic techniques or crystallization. Furthermore, the mild reaction temperature of 30°C suppresses thermal degradation pathways that often plague high-temperature heterocycle syntheses. The specific stoichiometry recommended in the patent—a molar ratio of palladium chloride to triphenylphosphine to sodium bicarbonate of 0.05:0.1:2—optimizes the catalytic turnover while maintaining a basic environment sufficient to drive the initial amidine formation without promoting hydrolysis of the sensitive imidoyl chloride. This precise balance of reagents ensures a clean reaction profile, facilitating the production of high-purity OLED material precursors or API intermediates that meet stringent regulatory specifications.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it accessible for both laboratory optimization and industrial manufacturing. The process begins by charging a reactor with the palladium catalyst system, the CO source components, and the base in an appropriate organic solvent. The specific choice of solvent, preferably tetrahydrofuran (THF), is critical for ensuring the complete dissolution of all reactants and maximizing reaction efficiency. Once the mixture is homogenized, the key building blocks—trifluoroethylimidoyl chloride, propargylamine, and the specific diaryl iodonium salt corresponding to the desired aryl substitution—are introduced. The reaction is then maintained at a constant 30°C for a period of 16 to 24 hours, allowing sufficient time for the catalytic cycle to reach completion. Following the reaction, the workup involves a straightforward filtration to remove inorganic salts, followed by silica gel mixing and column chromatography purification. This streamlined protocol minimizes unit operations and solvent consumption, directly contributing to process sustainability.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical novelty. The primary advantage lies in the significant simplification of the supply chain for raw materials. The starting materials, including aromatic amines, alkynylamines, and formic acid, are commodity chemicals available in bulk quantities from multiple global suppliers, reducing the risk of single-source dependency. This abundance translates directly into stabilized pricing and enhanced supply security for long-term projects. Moreover, the elimination of hazardous reagents like trifluorodiazoethane removes the need for specialized storage and handling infrastructure, thereby lowering capital expenditure (CAPEX) and operational expenditure (OPEX) associated with safety compliance. The ability to run the reaction at near-ambient temperature (30°C) further reduces energy consumption compared to processes requiring reflux or cryogenic conditions, aligning with modern green chemistry initiatives and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive catalysts and reagents. Palladium chloride, while a precious metal, is used in low loading (5 mol%), and the ligand triphenylphosphine is extremely cheap. More importantly, the avoidance of exotic fluorinating agents and the use of simple carboxylic acid derivatives as CO surrogates drastically lowers the material cost per kilogram of product. The high yields reported (up to 97%) mean less raw material waste and lower costs associated with recycling or disposing of unreacted starting materials. This efficiency allows for a more competitive pricing structure for the final pharmaceutical intermediates, providing a clear margin advantage for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply lines. The tolerance for various functional groups means that a single platform technology can be adapted to produce a wide library of analogues without needing to revalidate entirely new processes for each derivative. This flexibility allows suppliers to respond rapidly to changing R&D demands, shortening the timeline from custom synthesis requests to delivery. The use of stable solid reagents like diaryl iodonium salts also simplifies logistics, as they do not require the cold-chain shipping or inert atmosphere transport often needed for sensitive organometallic reagents, thus reducing lead times and shipping costs.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the mild exothermic nature of this reaction at 30°C makes it inherently safer and easier to scale from gram to tonnage levels. The generation of benign byproducts and the absence of heavy metal waste streams (beyond the recoverable palladium catalyst) simplify wastewater treatment and reduce the environmental footprint of the manufacturing site. This alignment with environmental, social, and governance (ESG) criteria is increasingly important for multinational corporations seeking compliant partners. The straightforward post-treatment involving filtration and chromatography ensures that the final product meets high purity standards with minimal processing steps, facilitating a smoother technology transfer to commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN111423381B, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their development pipelines.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method utilizes cheap and readily available starting materials like propargylamine and trifluoroethylimidoyl chloride, operating under mild conditions (30°C) with high substrate compatibility, avoiding the harsh conditions often required by direct trifluoromethylation reagents.
Q: What is the role of formic acid and acetic anhydride in this reaction system?
A: Formic acid and acetic anhydride serve as an in situ carbon monoxide (CO) source, facilitating the carbonylation step essential for forming the imidazole ring structure without requiring high-pressure CO gas cylinders.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is suitable for gram-level expansion and industrial large-scale production due to its simple operation, high reaction efficiency, and the use of stable, commercially available catalysts and solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the palladium-catalyzed carbonylation route described in CN111423381B and is fully prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce your overall project costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and delivering value through innovation.
